
Author: Kajal Gupta
Mentor: Dr. Emma Sarro
Abstract
Have any attempts been made to define food? Out of the three essentials in life, a meal is the complex chemical our bodies depend on for sustenance. Food is the third most vital component of life that sustains us, behind air and water. Food is obviously important and necessary. Our bodies need nutrients and energy to grow, be active and healthy, move, play, work, think, and learn. Healthy eating gives us these things. (Shriram, 22) [1]. Because each food or liquid has a certain nutrition, such as carbs, protein, vitamins, minerals, lipids, etc., which are crucial for our physical and mental development, foods are closely tied to our body, mental health, and social health.There is a strong bond between food and culture that is both practical and emotional. The food that was available to them served as the foundation for many aspects of different cultures. Making and consuming cuisine from one’s home country helps keep people who leave their cultural contexts linked. They also spread it throughout, and it fits nicely with the surrounding culture. [1,2 (Shriram, R. (22, 04 10). ACME COLLINS…) (Soken-Huberty, 21)]. The idea of a brain gut microbiome axis, which mediates bidirectional communication between the gut, its microbiome, and the neurological system, has been established, mostly based on findings from preclinical investigations. A variety of brain-gut illnesses may be influenced by changes in these interactions, according to the limited human data available.We all know that food has an impact on your physical health, but even the act of preparing it has significance. According to studies, confidence, focus, and happiness can all be boosted by cooking and baking. The justification is that cooking is a creative activity, and like other creative activities, it helps people feel happier and less stressed. It’s crucial to research how it contributes to or treats mental disease.The main inquiry of this study, which is supported by pertinent evidence, is how certain everyday, widely consumed foods, such as coffee, tea, nuts, vegetables, or various dietary patterns, such as intermittent fasting or probiotic plant-based diets, affect a person’s overall lifestyle and how they may affect conditions like schizophrenia, which are not yet clearly linked, are unknown.[3,4, (Quddusi, 2018…. (Osadchiy, 2019)]
Keywords : microbiology, diagnosis, microbiome, genetics
Introduction
Have you ever wondered whether food could be more than just a fundamental necessity for survival? Everyone is aware of the influence food has on a person’s life, for example, how eating a low-carb diet makes one thinner, how eating a lot of protein makes one muscular, or how vitamin consumption affects how well one’s skin and hair appear. All of these physical effects, or to put it another way, discernible changes, are today’s concerns of every person when it comes to food. Some modifications, nevertheless, are not readily apparent. It’s always simpler to connect food with physical wellbeing, not giving a second consideration to how food can affect mental wellbeing, which is another factor in an individual’s general wellbeing.
Mental health
Interestingly, one of the most common things to do when one is unhappy is to eat sweets or chocolates. A popular adage is “Stressed is backwards transcribed as desserts,” but one shouldn’t stop there. One could also consider why and what motivates people to seek for something sweet when they experience stressful or depressing emotions.
Bidirectional communication of brain and gut, The complex bidirectional relationship between the gut and the brain involves systems like the endocrine, immune, autonomic, and enteric neurological systems. Additionally, there is increasing evidence that the microbiome and the chemicals made by the microbiota play a part in regulating these connections; however, the mechanisms underlying these effects are still being worked out. The interactions between the microbiota and gut peptides are anticipated to have a significant impact on the modulation of gut-brain signaling.[5Carabotti, M. S. (2015). Carabotti, M…]
Have any attempts been made to define food? Out of the three essentials in life, a meal is the complex chemical our bodies depend on for sustenance. Food is the third most vital component of life that sustains us, behind air and water. Food is obviously important and necessary. Our bodies need nutrients and energy to grow, be active and healthy, move, play, work, think, and learn. Healthy eating gives us these things. (Shriram, 22) [1]. Because each food or liquid has a certain nutrition, such as carbs, protein, vitamins, minerals, lipids, etc., which are crucial for our physical and mental development, foods are closely tied to our body, mental health, and social health.There is a strong bond between food and culture that is both practical and emotional. The food that was available to them served as the foundation for many aspects of different cultures. Making and consuming cuisine from one’s home country helps keep people who leave their cultural contexts linked. They also spread it throughout, and it fits nicely with the surrounding culture. [1,2 (Shriram, R. (22, 04 10). ACME COLLINS…) (Soken-Huberty, 21)]. The idea of a brain gut microbiome axis, which mediates bidirectional communication between the gut, its microbiome, and the neurological system, has been established, mostly based on findings from preclinical investigations. A variety of brain-gut illnesses may be influenced by changes in these interactions, according to the limited human data available.We all know that food has an impact on your physical health, but even the act of preparing it has significance. According to studies, confidence, focus, and happiness can all be boosted by cooking and baking. The justification is that cooking is a creative activity, and like other creative activities, it helps people feel happier and less stressed. It’s crucial to research how it contributes to or treats mental disease.The main inquiry of this study, which is supported by pertinent evidence, is how certain everyday, widely consumed foods, such as coffee, tea, nuts, vegetables, or various dietary patterns, such as intermittent fasting or probiotic plant-based diets, affect a person’s overall lifestyle and how they may affect conditions like schizophrenia, which are not yet clearly linked, are unknown.[3,4, (Quddusi, 2018…. (Osadchiy, 2019)]
Part of the brain-gut mechanism involves the complexity of the gastrointestinal (GI) tract. The GI tract contains specialized enteroendocrine cells (EECs) that release more than 20 signaling chemicals that can communicate with the brain and carry out critical metabolic and endocrine functions [6 (J., Gastrointestinal hormones and the dialogue between gut and brain., 2014) ]. It is now clear that EECs, which make up less than 1% of the total population of gut epithelium cells, are regulated by the gut microbiota [7,8,9 (J., Enteroendocrine cell signalling via the vagus nerve., 2013) (Cani, (2013)) (Cohen, 2017)Cohen, L. J.-F. (2017). Commensal…], which as a whole describes the population of non-human bacterial organisms. The diversity and make-up of enteric bacteria directly affects the release of gut peptides like glucagon-like peptide-1 (GLP-1), peptide YY (PYY), cholecystokinin (CCK), corticotropin-releasing factor (CRF), oxytocin, and ghrelin [6,10,11,12 (J., Gastrointestinal hormones and the dialogue between gut and brain., 2014)J., D. G. (2014). Gastrointestinal hormones and the…, (Latorre, 2016) (Psichas, (2015).) (Gribble, 2016]. To understand the mechanism of gut peptides through the brain- gut axis refer (figure 1) .We still don’t fully understand the mechanisms underlying interactions between the EEC and the microbiota.
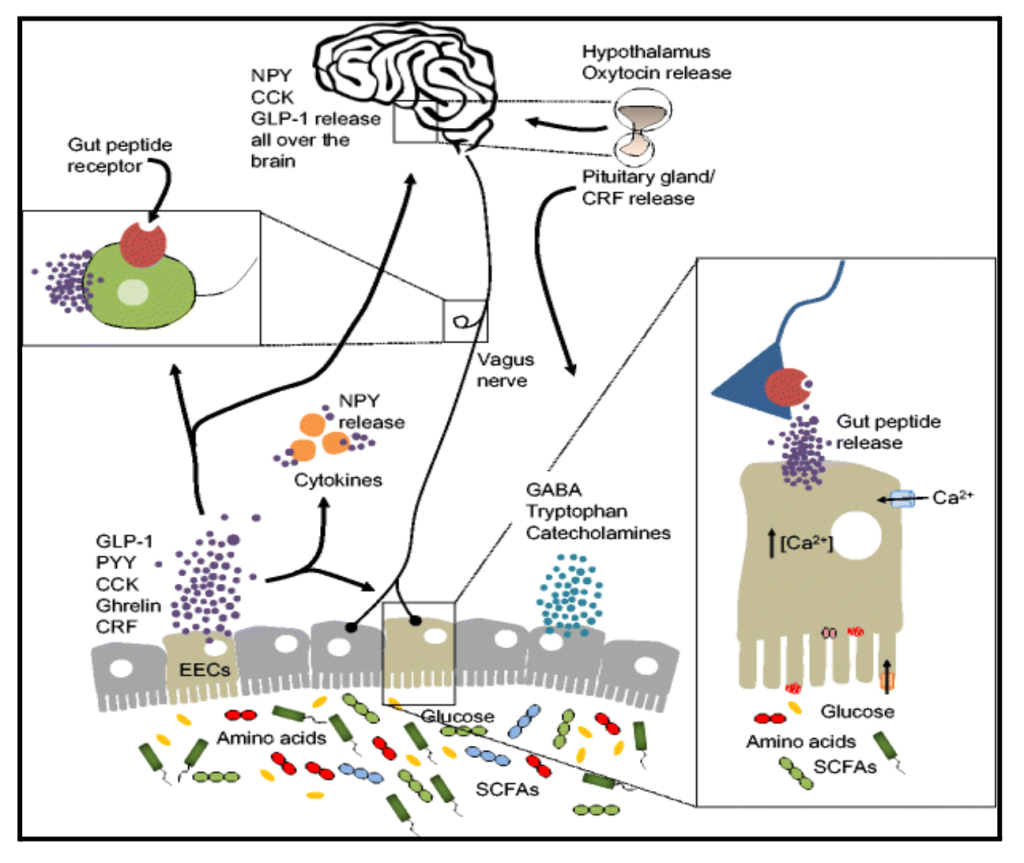
Note. There are numerous mechanisms through which the gut microbiota may signal the brain to control physiological processes. These include the release of gut peptides by enteroendocrine cells (EECs) where they activate cognate receptors of the immune system and on vagus terminals in the gut. Gut peptides such as neuropeptide Y (NPY) can also be released by cytokines under immune stimulation. There have been numerous reports of alterations in the gut microbiota in neuropsychiatric conditions, where gut peptides may play a key signaling role. Only a few examples of gut–brain pathway and gut peptides are represented in this figure.
CCK = cholecystokinin; GLP-1 = glucagon-like peptide; CRF = corticotropin-releasing factor; PYY = peptide YY; GABA = γ-aminobutyric acid; SCFA = short-chain fatty acid. [5 (Carabotti, 2015)]
See Table 1 for a summary of many compounds produced by bacteria that have been found to have an effect on the brain.(Table 1).
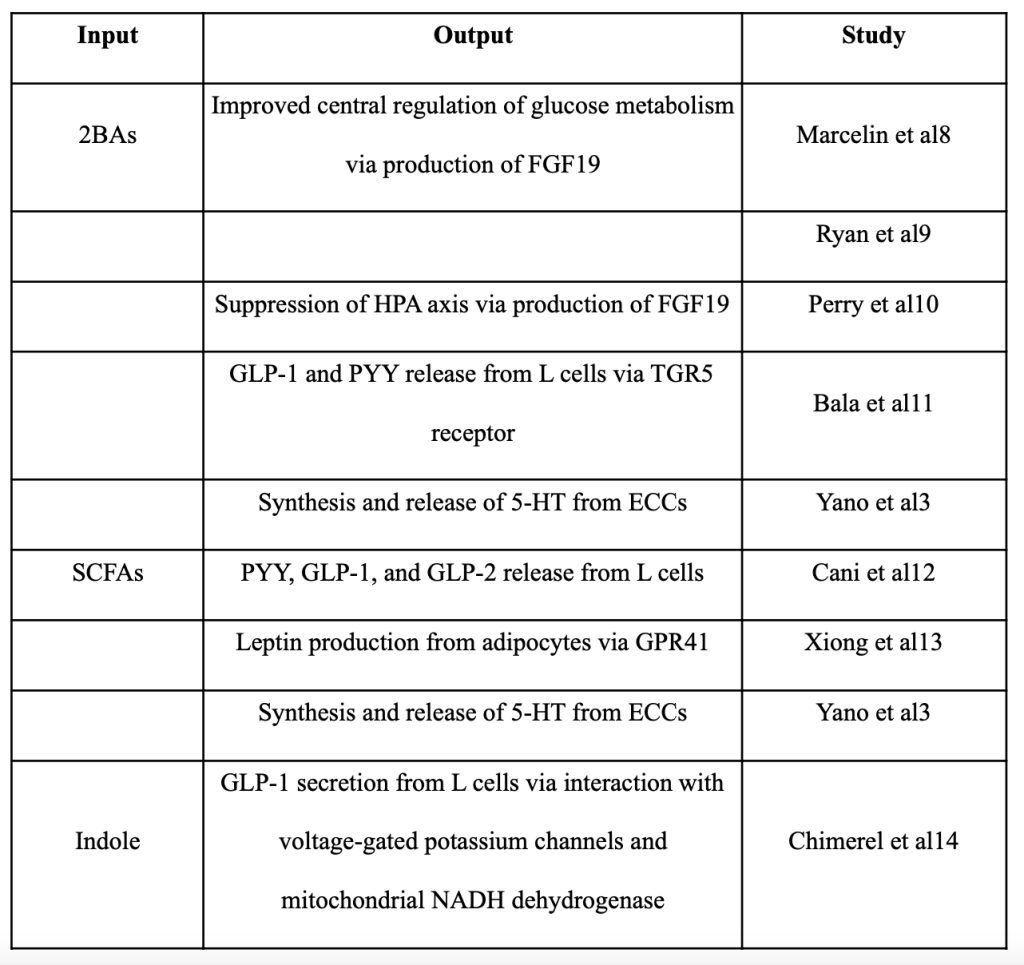

In addition to the signaling molecules mentioned above, the gut and the brain are continually communicating with one another. Such communication may have an impact on an individual’s perception of visceral events including nausea, satiety, and pain, and it may be crucial in mediating physiological effects ranging from GI function to the brain and behavior. In addition to the information traveling to the brain, there are brain-based signals that travel in the other direction. For example, GI secretions and motility are impacted by stressful circumstances. The therapeutic relevance of such bidirectional communication and alterations in the gut flora are currently the subject of investigations. GI tract sensory information that is transformed into neural, hormonal, and immunological signals may be the beginning of the bidirectional gut-brain link [13 (Bellono, (2017))]. These signals can carry information to the brain singly or in combination (CNS). The current study touches on the immunological and neurological pathways as necessary.
The gut-brain axis (GBA), which links the brain’s emotional and cognitive centers to the intestine’s peripheral functions, serves as a conduit for communication between the central (brain and spinal cord) and enteric (gut) neural systems. Recent advances in science have emphasized the importance of the gut microflora in influencing these interactions. This interaction between microbiota and GBA appears to be bidirectional, with signals traveling both ways from the gut microbiota to the brain and from the brain to the gut microbiota via neurological, endocrine, immunological, and humoral links. [4 (Osadchiy, 2019)]
Understanding the crosstalk between the gut and the brain has revealed a complicated communication system that ensures the proper maintenance of gastrointestinal homeostasis. Additionally, several effects on mood, motivation, and higher cognitive abilities are anticipated from this system. [14 (Rhee SH, 2009;)]. In addition to connecting the brain’s affective and cognitive centers to peripheral intestinal activities, its duty is to monitor and integrate gut events. Mechanisms involved in this process include immune activation, intestinal permeability, the enteric reflex, and entero-endocrine signaling. The mechanisms underlying GBA communications involve neuro-immuno-endocrine mediators. Figure 2 represents a summary of this relationship, and Figure 3 .[4 (Osadchiy, 2019)]
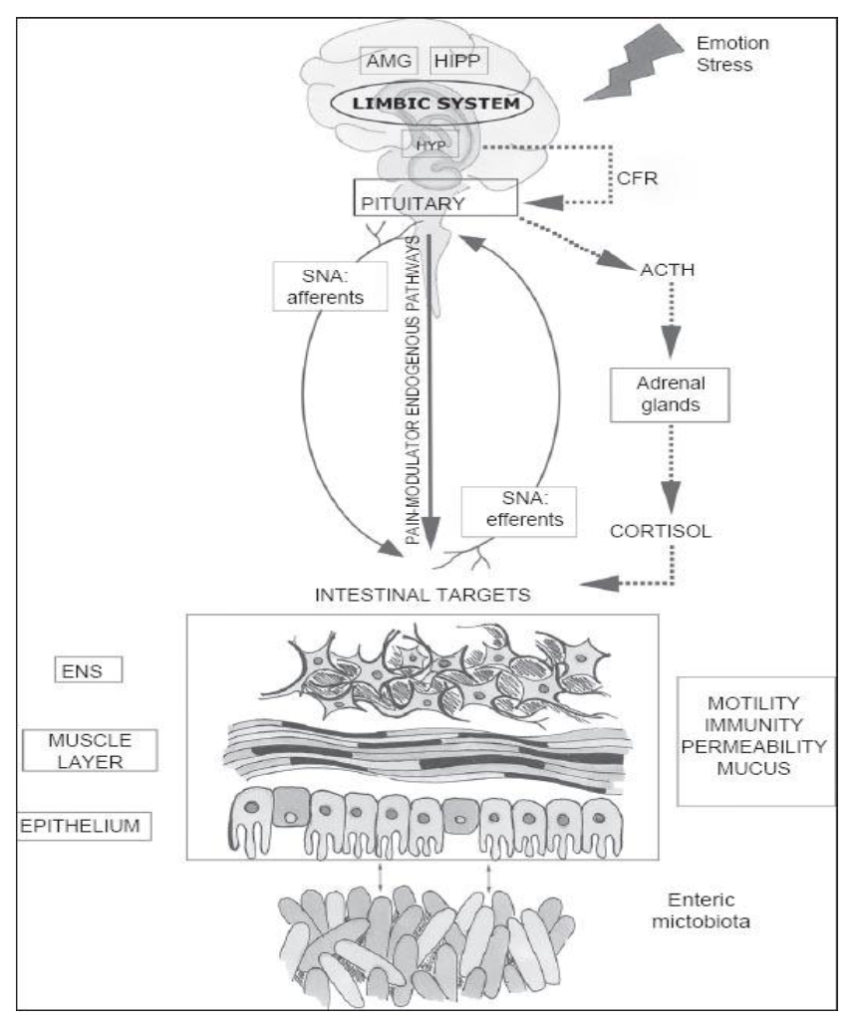

Eating patterns
Our eating patterns can also impact this relationship.[18 (Simeonova D, 2018)] For example. stress can cause a “leaky gut” [17,19 (Jiang H, 2015) (Bonaz BL, 2013)]by increasing the permeability of the intestinal barrier. The mucosal membrane may become permeable to bacteria and their byproducts as a result of this loss of integrity.[19,20 (Bonaz BL, 2013) (Friebe A, 2011)] In many mental health diseases, including Alzheimer’s and schizophrenia[20,21 (Friebe A, 2011) (Köhler CA, 2016;)], this bacterial translocation can result in autointoxication, which fuels the chronic inflammatory state.[15, 16 (Alagiakrishnan, (2020) ) (Souza DG, 2004;)]
In actuality, how we relate to food is a significant factor. A recent line of research has focused on encouraging conscious food selection, increasing awareness of the distinctions between physical and psychological hunger, observing the symptoms of satiety, and eating healthfully in response to all of these cues. In this current body of literature, the “mindful eating” (ME) movement places an emphasis on being mindful of the present moment while eating, observing the impact of food on the senses, and accounting for one’s bodily and emotional reactions to the eating experience.The imbalance of the structure and operation of the gut intestinal microbiota is known as microbial dysbiosis. It is a widespread issue in the modern period and is brought on by bacterial infections, dietary changes, and antibiotics. Recent studies have shown that intestinal illnesses like [Irritable bowel disorder.] IBD, irritable bowel syndrome (IBS), and celiac disease are related to dysbiosis of the gut microbiota. Through competition for nutrients and space, beneficial bacteria in the digestive tract prevent harmful bacteria from infiltrating and growing. Intriguingly, probiotics are one of the most important treatments for reestablishing the gut’s microbial balance and preventing infections in patients after antibiotic therapy, which results in dysbiosis of the gut microbiota.[23, 24, (Morillo Sarto, 2019) (Gerald J Holtmann, 2016)]
Link to cognitive disorders
With the development of genomic technologies, schizophrenia (SZ) may now be studied in its potential relationship to gut microbiome, which can yield a wealth of information for the potential diagnosis and prognosis of SZ. Diverse microbial taxa drove community splitting in each of the previous research of the gut microbiome in SZ patients, which confirmed that the species structure inside the stomach of SZ patients differs from that of normal control subjects (NCs), and a number of these research findings also concentrated on the association between various gut microbiota and clinical characteristics [26,27,28, (Nguyen T. T., 2019) (Schwarz E, 2018;),, (Li S. Z., 2020)] Other neuropsychiatric illnesses, in addition to SZ, like depression[29], bipolar disorder [30 (Hu, 2019)], autism[31 (Tomova, 2015)], Alzheimer’s disease [32 (Cattaneo, 2017)], and Parkinson’s disease[33 (Caputi, 2018)], have been linked to a dysregulated MGB axis.[25, 34, 35 (Li S. S., 2021) (Desbonnet, 2014) (Sampson, 2016) ] (organize the citations)
One potential source for this relationship is Brain-derived neurotrophic factor (BDNF) and glial cell line-derived neurotrophic factor (GDNF), which can both influence neural development and brain plasticity[36 (Sudo, 2004)], and are just two examples of the neurotrophic factors whose expression the gut microbiota can regulate. Recently, new information on the MGB axis in SZ patients has been published. According to Nguyen et al.[37 (Nguyen T. T., 2018)], SZ may be linked to gastrointestinal inflammation. Additionally, SZ patients frequently have gastrointestinal and digestive disorders[38 (Saha, 2007)]. The connection between the gut microbiome and brain shape and function, however, has received little research attention.
Impact of specific food items
Finally, there is broad agreement that plant-based eating regimens like the Mediterranean, DASH(Dietary Approaches to Stop Hypertension), and MIND diets (Mediterranean-DASH Intervention for Neurodegenerative Delay) enhance general cognitive function and prevent age related cognitive decline (ARCD). Numerous plant foods, including berries high in polyphenols, fruit juices, coffee, tea, cocoa, and nuts, have a positive effect on cognitive health. When it comes to frontal executive functions like attention, processing speed, and memory encoding, consolidation, and retrieval, polyphenols appear to have the strongest protective effects. Possible processes underpinning the neurocognitive advantages include better neurogenesis, decreased oxidative stress and neuroinflammation, increased cerebral blood flow, and neuroplasticity [40,41,42 (Joris PJ, 2018) (Lamport DJ, 2015;) (Flanagan E, 2018;.)]. In addition to polyphenols, n-3 PUFAs and other antioxidant micronutrients may collaborate to improve cognitive wellbeing.[39, (Sujatha Rajaram, Issue Supplement_4, November 2019)]
In this review, we will concentrate on dietary compounds that directly interact with gut microorganisms and travel through the gut-brain axis to the brain, and we’ll talk about how that affects host physiology, gut microbial ecology, and health. Even thousands of years later, Hippocrates’ dictum that “Let food be thy medicine and medicine be thy food” is still very much applicable and requires thought about how diet might be utilized to influence brain ecology to improve health.
Methods
For this review, a literature research was carried out using primarily the following databases: NCBI, PubMed, ScienceDirect , Google Scholar search engine to collect all of the literature cited. In our search, the parameters included studies published in the last 10 years, and available in English. Keywords used to search included: types of food, cognitive disorders , gut microbiota , brain-gut axis ,impact of food on mental illness, dietary patterns ,cultural differences . The studies gathered in our review included only patients with AD , dementia, SZ and some common gut-brain illnesses . The patients were in the age bracket of above 60 years of age and even childrens .
We relied on the above-mentioned search engines for assistance and data. We wanted to include review papers , meta – analysis with experimental evidence of the impact of food on the cognitive abilities of normal persons’ brains. In the end we included a total of roughly 50 articles.
The purpose of this narrative review is to investigate the effectiveness of diet in contribution and improvement of various cognitive disorders , so many articles contained these interventions for our data collection. A literature research was carried out using the following databases: NCBI, PubMed, ScienceDirect, Google Scholar
All clinical trials were used to create a more comprehensive review. These included interventional trials randomized controlled trials, observational trials, case-control studies, systematic reviews etc. Furthermore, this study focused majorly on the types of food and neurochem they contains as well the gut microbiome : thus, the type used in the intervention comparative studies for different geographical regions and ages to study the impact of different food and their different outcomes over a person’s cognitive health was another important factor when choosing articles.
Results
In order to form a link between the foods we eat and their possible link to brain health via the gut axis, we first examined how different food items compared to levels of fuel, d-glutamate,In addition to having a significant impact on cognition, behavior, and psychiatry, molecular chirality has been shown to influence protein folding, neuronal proliferation, and brain functional laterality [44 (Dyakin, Justin Lucas Dyakina-Fagnano, Posner, & Vadasz, 2017, )]. Enantiomers may differ significantly from one another due to differing pharmacologic and pharmacokinetic features, according to previous research [45 (Baker, Prior, & Coutts, 2002,)]. The notion that d-type amino acids are new neurotransmitters is supported by a recent research discovery [46Mothet, J., & Snyder, S. (2012,…]. Experiments on d-glutamate are scarce, and it is unclear how they affect neurocognitive function. A pilot human investigation revealed that d-glutamate levels are related to cognitive skills in SZ or AD patients [49 (Lin, Yang, & Lane, 2019, ) ], while animal studies have found greater d-glutamate levels in particular brain regions [47,48 (Mangas, et al., 2007, ) (Kawase, et al., 2017, )] This can also be seen in Table 2.
The brain-gut-microbiota axis may significantly contribute to neurodegeneration,Clinical appearance of neurodegenerative disorders is determined by the increasing malfunctioning of particular populations of neurons. The characteristic of many neurodegenerative proteinopathies, extracellular and intracellular accumulation of misfolded proteins, is linked to neuronal death.[43 (Chang, (2020)) (Dyakin, Justin Lucas Dyakina-Fagnano, Posner, & Vadasz, 2017, )]
Furthermore, glutamate signaling disruption, the brain’s main excitatory neurotransmitter, has a role in poor learning and memory. In individuals with neuropsychiatric diseases, changes in the gut microbiota and metabolite composition, particularly glutamate, have been observed. Two-keto-glutaramic acid, a glutamate metabolite, is lowered by gut microbiota Bacteroides vulgatus and Campylobacter jejuni. Additionally, the gut bacteria Corynebacterium glutamicum, Brevibacterium lactofermentum, and Brevibacterium avium can convert l-glutamate to d-glutamate using glutamate racemase.
Additionally, benzoate, a DAAO (d-amino acid oxidases) inhibitor, can raise d-serine levels, which in turn promotes NMDAR activation. Previous pilot studies have discovered that benzoate may help schizophrenia patients’ cognition. The movement of glutamate is controlled by the blood-brain barrier (BBB), which is the endothelial lining of the brain capillaries. Brain-to-blood glutamate efflux can be mediated by brain capillary endothelial cells [51,50 (Helms, Nielsen, Waagepetersen, & Brodin, 2017, ) (Hawkins & Vina, 2016, )]. The blood contains glutamate concentrations that are 5 to 50 times lower than those in the brain, preventing glutamate from entering the brain [52 (Bai & Zhou, 2017, )]. The two membranes that make up the BBB are luminal (blood facing) and abluminal (brain facing), and they complement one another in their functions. Only the luminal membranes have facilitative glutamate transport, whereas the abluminal membrane has a glutamate transport mechanism that is Na+-dependent. Transmembrane proteins known as glutamate transporters are produced by the translation of members of the solute carrier (SLC) gene families SCL 1, SLC 7, SLC 17, and SLC 25 [53 (Fotiadis, Kanai, & Palacin, 2013, )].
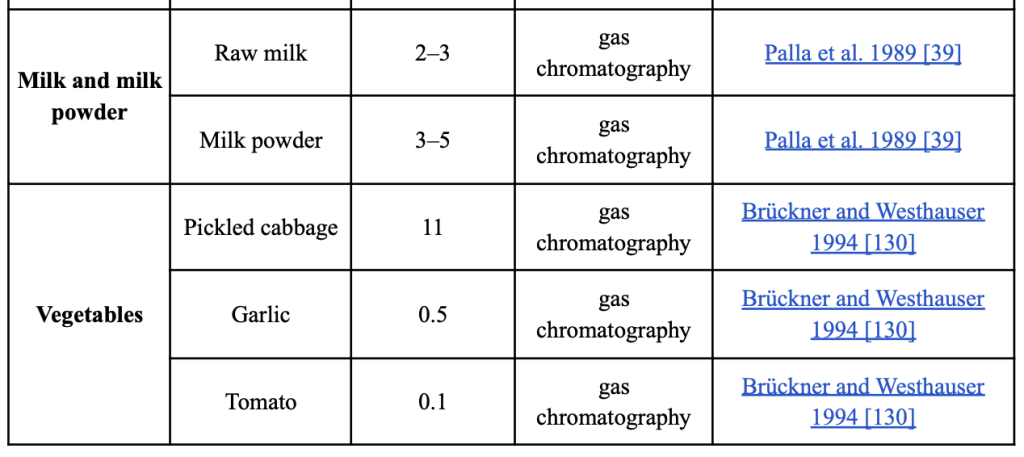
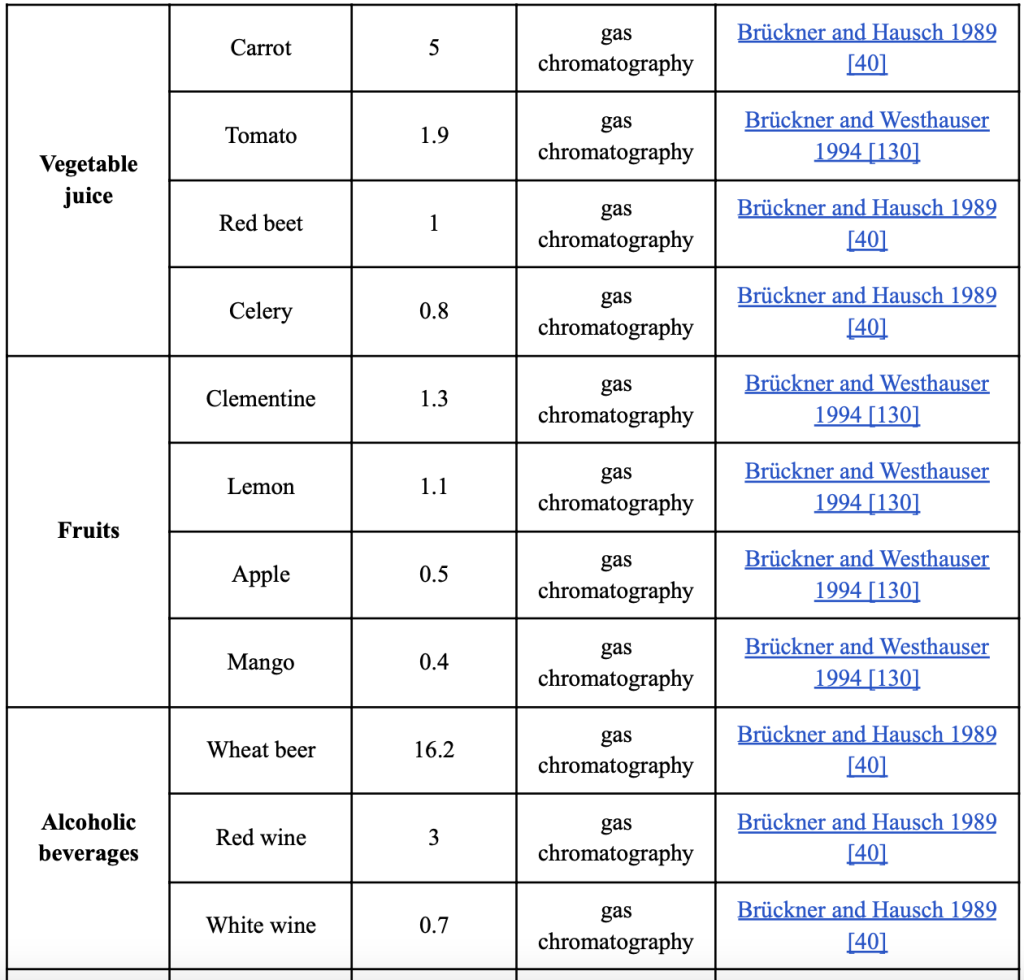
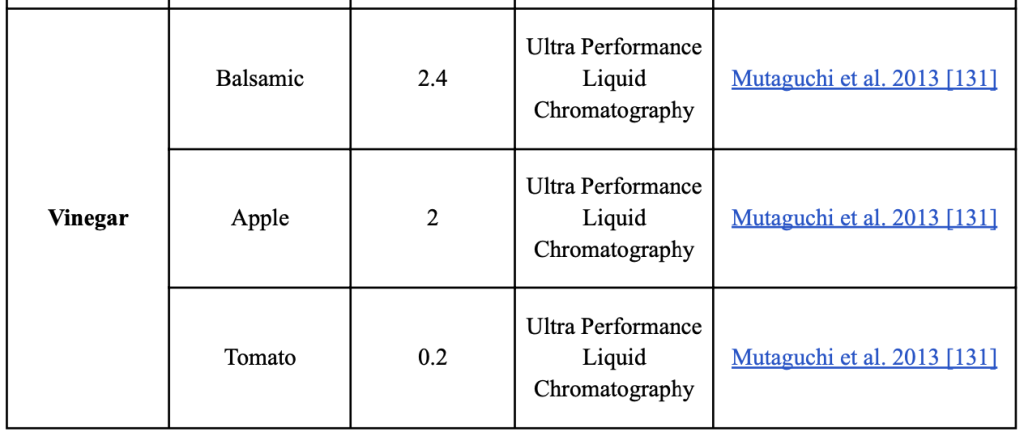
Human glutamate metabolism has been extensively studied in earlier studies. Selective transporters, such as excitatory amino acid carrier C1 (EAAC1) on the apical membrane of enterocytes, are able to absorb the most amino acids in the gastrointestinal tract after ingesting foods containing glutamate [54 (Burrin & Stoll, 2009, )]. Approximately 75%-96% of enteral glutamate is transported to the splanchnic arteries during the first-pass effect. Additionally, >80% of this glutamate is transformed into energy to sustain intestinal function [56,55 (Janeczko, Stoll, Chang, Guan, & Burrin, 2007, ) (Hays, Ordonez, Burrin, & Sunehag, 2007, )]. The synthesis of gut proteins may potentially be influenced by glutamate. They participate in a number of metabolic pathways, including the creation of important amino acids like arginine and proline, citrulline, and the antioxidant glutathione [57Reeds, P., Burrin, D., Stoll, B.,…]. They are also transformed into carbon and nitrogen donors. Additionally, along with l-form amino acids, d-form amino acids have recently been discovered to exist in a variety of foods and beverages, including milk, cheese, fish, vegetables, fruits, alcoholic beverages, vinegar, ham/meat, coffee, and milk powder [58Marcone, G., Rosini, E., Crespi, E.,…]. The reported proportion of d-glutamate in relation to the total amounts of amino acids is summarized in [Table 2]. Fermented foods contain significant amounts of d-glutamate. 27.4% of instant coffee [59,60 (Palla, Marchelli, Dossena, & Casnati, 1989, ) (Brückner & Hausch, Detectio0n of free D-amino acids in food by chiral phase capillary gas chromatography. , 1989, )] and 32%-41% of roasted coffee, respectively, contain d-glutamate. Yogurt has 12.4% d-glutamate, Yakult has 24.2%, Parmigiano Reggiano has 15%, and so on [62,63,61 (Jin, Miyahara, Oe, & Toyo’oka, 1999, ) (Marchelli, et al., 2007; ) (Brückner & Hausch, D-amino acids in dairy products: Detection, origin and nutritional aspects. I. Milk, fermented milk, fresh cheese and acid curd cheese. , 1990, )]. Varied fermentation techniques could result in different amounts of d-amino acids being produced. Different types of D-amino acids are produced by lactic fermentation using lactic acid bacteria [64 (Kobayashi, 2019, )For instance, the glutamate racemase in Lactobacillus plantarum changes l-glutamate into d-glutamate.[65 (Bohmer, Dautel, Eisele, & Fischer, 2013, )]
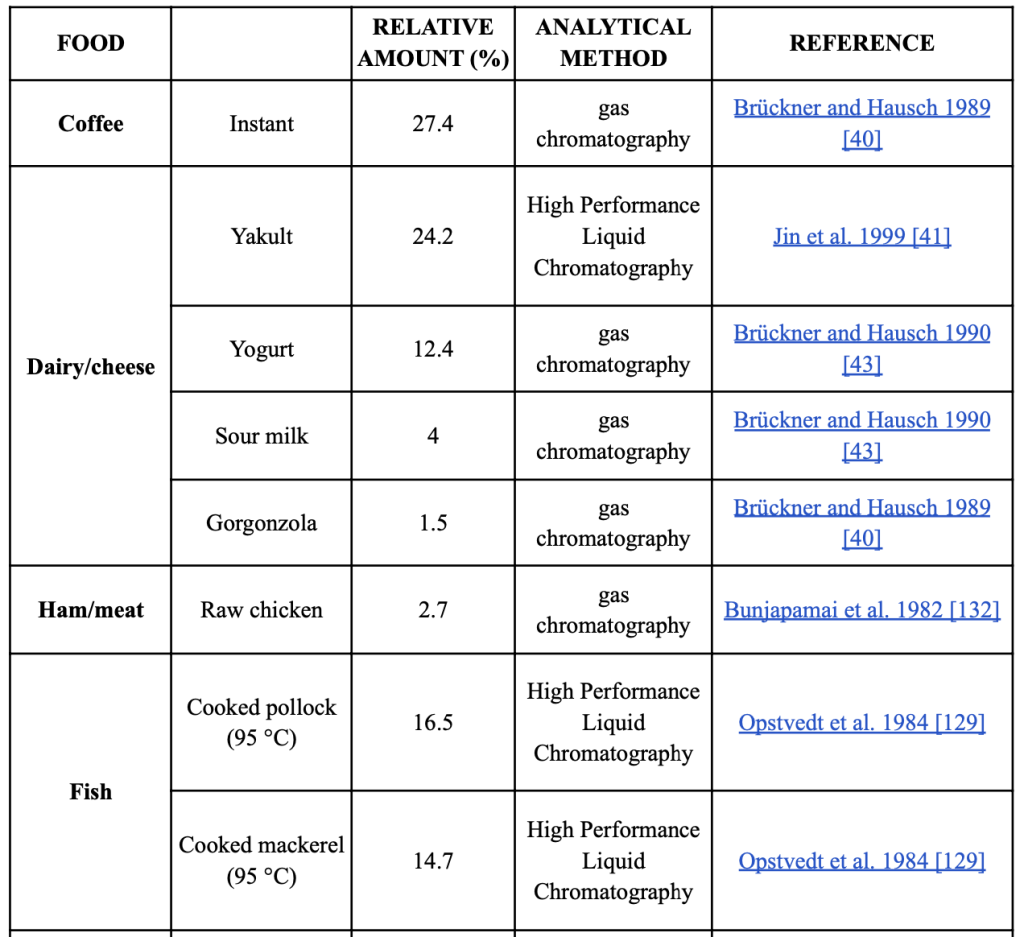

In order to understand whether there is a relationship between cognition based on eating specific food types, we describe several types of food below. This can also be seen in Table 2
Phenol-rich berries and fruit juices
Polyphenols, such as found in grape, citrus fruits – oranges , promgerant , cherries, cocoa etc, have been shown to have positive impacts on brain health in studies [66 (Gildawie KR, 2018;)], and there is growing evidence linking polyphenol consumption to slowed cognitive aging in older adults [67 (Valls-Pedret C, 2012)]. Review of studies on fruit juices high in polyphenols can be found in Table [3]. Despite the limited amount of research, fruit drinks, such as orange, grape, and pomegranate juice, seem to have a good impact on cognition. In healthy older people, consumption of high-flavanone orange juice (OJ) increased global cognition as measured by the mean z-score of numerous measures of executive function and episodic memory when compared to a low-flavanone control (500 mL/d for 8 wk) [68 (Kean RJ, 2015;1)]. Notably, effects were primarily seen in immediate recall on tests of episodic memory, which frequently reflects frontal executive components of attention and memory encoding.
Among beverages high in antioxidants, pomegranate juice has the highest content of phenolics and the most antioxidant activity, followed by red wine, Concord grape juice, and blueberry juice [69 (Harnly JM, 2006;)]. Pomegranate juice contains a large amount of anthocyanins and hydrolysable tannins, such as glycosides of ellagic acid. In comparison to the placebo group, older persons who took pomegranate juice for four weeks showed enhanced encoding and retrieval on a test of verbal memory [70 (Bookheimer SY, 2013;)]. These improvements were also accompanied by increases in brain activity that were specific to the verbal and visual tasks. Pomegranate polyphenols also showed to improve memory function by boosting cerebral blood flow during certain tasks.
In four small RCTs(Randomized Controlled Trials), older persons who regularly consumed grapes (100 percent Concord grape juice or an active grape formulation made from freeze-dried grape powder from California grapes) were compared to those who consumed a placebo. In older persons with early memory deterioration but without dementia, daily ingestion of Concord grape juice (6.3–7.8 mL/kg body weight for 16 weeks) improved Verbal Learning Test results as compared to placebo [71 (Krikorian R N. T.-H., 2010;)] scores.What’s interesting about this is that there was no measurable difference after 16 weeks of daily Concord grape juice consumption suggesting There was no difference in learning and retention scores, but therapy resulted in less semantic interference on memory tasks (P = 0.004) and more right hemisphere activity on fMRI.[40 (Joris PJ, 2018)]
Overall, citrus fruit juices, pomegranate juice, grape juice, and cherry juice, all with large amounts of polyphenols of different subtypes, appear to exert brain-protective effects and improve global or domain-specific cognitive function in as little as 4 weeks of consumption. Based on the above studies, acute benefits after a single dose are unlikely and longer-term daily consumption could be necessary to achieve observable effects on cognition. Further, benefits might be less relevant for younger adults and more pronounced in older adults at risk for either age-related or pathological cognitive decline. Specifically, there appears to be most benefit in frontal-executive functions including attention and memory encoding and retrieval.
Additionally, flavonoids like catechin and epicatechin, kaempferol and myricetin, as well as the anthocyanins delphinidin and petunidin, are all abundant in berries. Through a number of hypothesized mechanisms, including the suppression of microglia-mediated inflammation and the reduction of vascular risk through a decrease in blood pressure and oxidative stress, which is aided in part by neuronal and inducible nitric oxide production, these flavonoids are thought to be neuroprotective and slow brain aging [66,73, 72 (Gildawie KR, 2018;) (Potì, (2019)) (Spagnuolo C, 2018)].
Human research has backed the use of berries to enhance cognitive well-being, following data from animal studies confirming the importance of berry-rich diets in motor performance, working memory, and enhanced neurogenesis [76,75 (Galli RL, 2016) (Shukkit-Hale B, 2015;)]. [74,77-79 (Nones, 2011)) (Devore EE, 2012) (Travica N, 2019, ) (Kent K, 2017;) ]. Strawberries and blueberries dramatically slowed the deterioration of elderly women’s language comprehension and global cognition, [69 (Kean RJ, 2015;1)]. The impacts of reinforcing with various blueberry formulations on cognition were examined in three RCTs, and a mixed-berry beverage containing blueberries, blackcurrants, elderberries, lingonberries, strawberries, and tomatoes was used in a fourth [80-84 (Whyte AR, . 2018;.) (Miller MG, 2018;) (Bowtell JL, 2017;) (Nilsson A, 2017;) (Krikorian R S. M.-T.-H., . 2010)]. (Table 3). In healthy senior people, reductions in shifting inaccuracies on a task-switching test and repetition errors on a verbal learning test were seen after consuming a freeze-dried blueberry supplement (equal to about 150 g fresh blueberries) for 90 days[81 (Miller MG, 2018;)]. Similarly, robust seniors’ efficiency in visual recall and episodic recognition memory was improved after consuming blueberry concentrate for three months [82 (Bowtell JL, 2017;)]. After just 5 weeks, blueberries improved working memory in healthy older subjects when combined with other fruits and berries, but no changes were seen in the areas of selective attention or psychomotor reaction time [84 (Krikorian R S. M.-T.-H., . 2010) ]. Elderly subjects with early cognitive impairment or more severe cognitive complaints showed improved verbal memory function with blueberry supplementation [80,84-86 (Whyte AR, . 2018;.)(Krikorian R S. M.-T.-H., . 2010) (McNamara RK, 2018;) (Bensalem J, 2019;)]. According to brain imaging studies, increased brain perfusion and activation of brain regions relevant to cognitive function appear to go hand in hand with the beneficial effects of blueberry supplementation on cognition domains [82 (Miller MG, 2018;)].
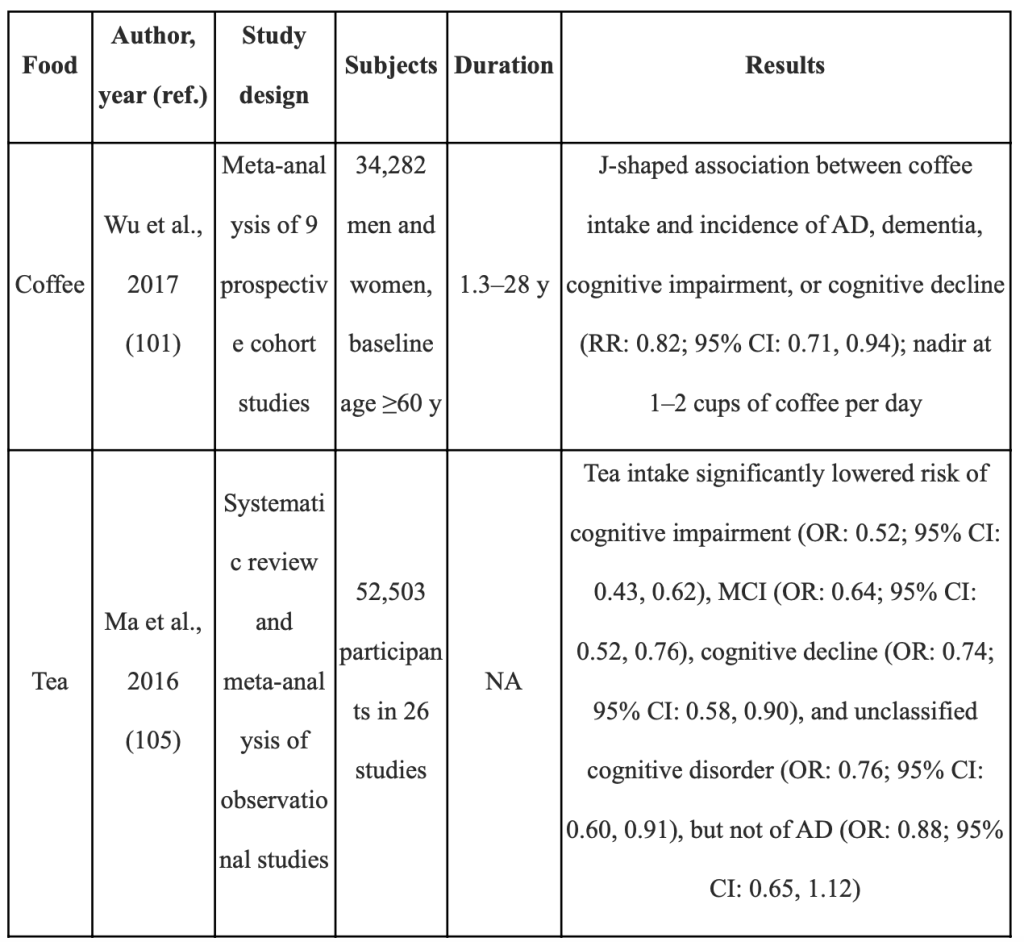
Both tea and coffee, which are well-known sources of caffeine, are also great sources of polyphenols. Coffee includes significant levels of phenolic acids, including some catechols and the hydroxycinnamic acids 4- and 5-caffeoylquinic acids (19 and 43 mg/mL, respectively). Catechins, epicatechins, and procyanidins are the main flavonoids found in green tea, while hydroxybenzoic and hydroxycinnamic acids and 5-caffeoylquinic acid are found in smaller concentrations. Green tea and coffee are frequently the most significant sources of polyphenols in diets [88,87 (Zamora-Ros R, 2016;) (Burkholder-Cooley N, 2016;)]. For example, in a prospective cohort study of seniors with cognitive stability (aged 69 to 86 years), participants who consumed coffee had a lower risk of showing decline on two cognitive tests over the course of three years[89 (Haller S, 2018;)]. Additionally, white matter preservation was better in cognitively stable participants who consumed coffee in the second and third tertiles. Green tea catechols, particularly epigallocatechin gallate, but also epigallocatechin, epicatechin, rutin, and L-theanine, may play a role in mitochondrial dysfunction and the prevention of amyloid formation [90 (Polito CA, 2018;)]. Together, regular consumption of tea and coffee may reduce the likelihood of developing cognitive problems, however the relationship is J-shaped for coffee and linear for green tea.
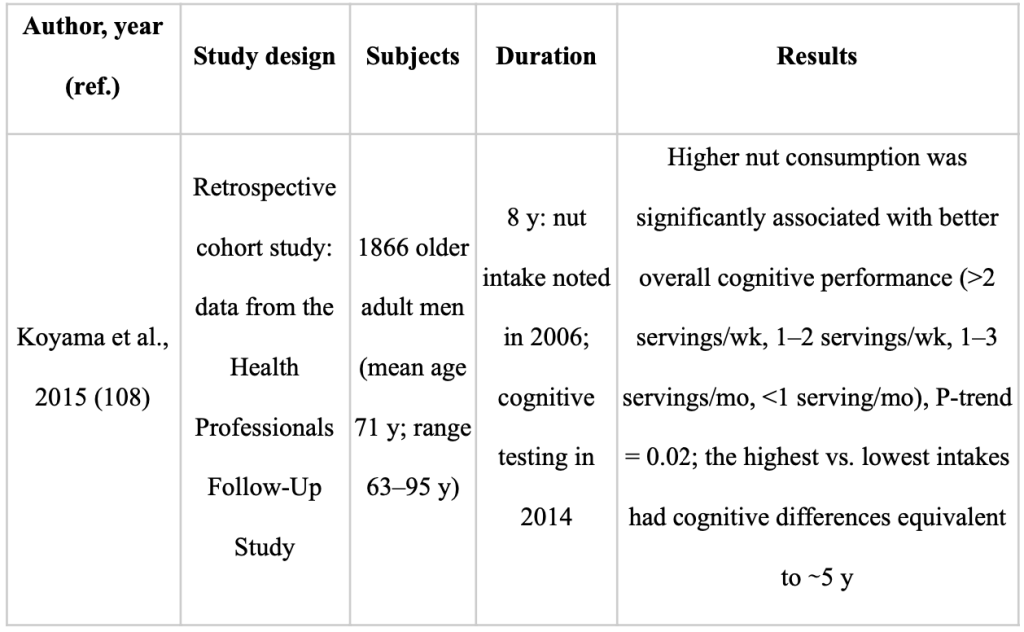
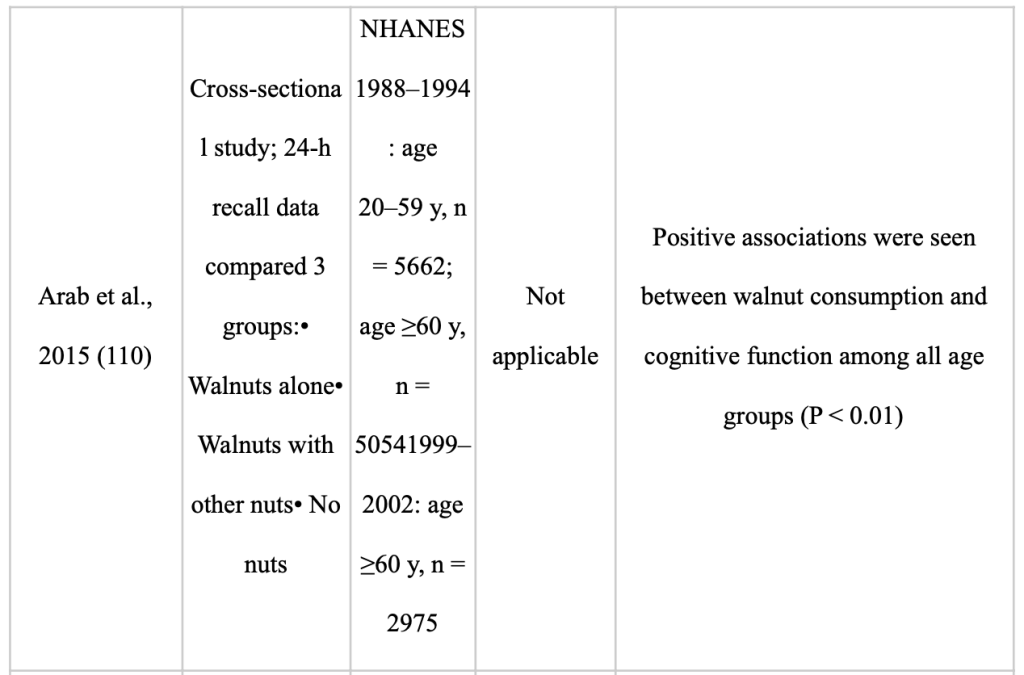
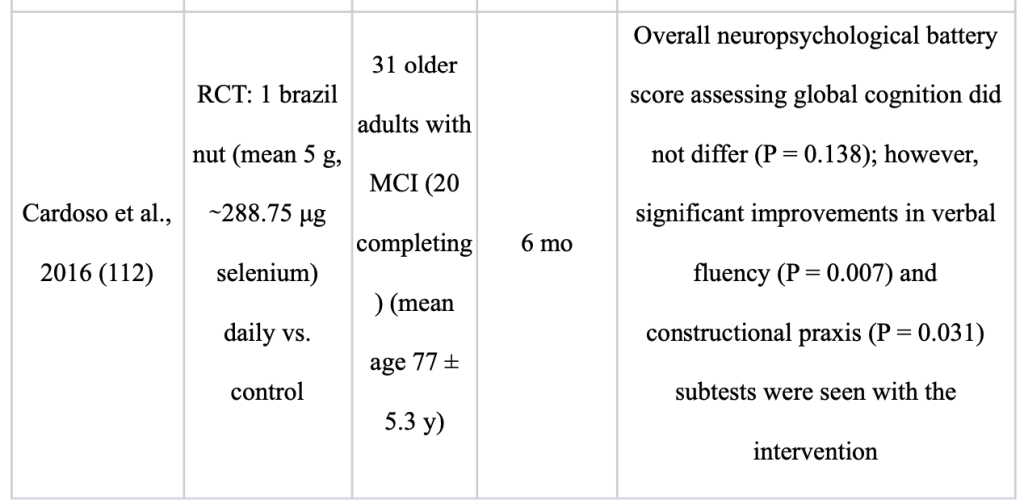
Last but not least, tree nuts and legumes are likewise abundant in polyphenols, albeit there haven’t been many studies on their effects on human cognition [Table 4 ]. This evaluation does not include research that examines the relationship between nut consumption and cognition in younger individuals or studies that take into account postprandial effects. All nuts, but walnuts stand out for their nutrient-rich matrix of polyunsaturated fats, including both linoleic and -linolenic acid and antioxidant polyphenols that may influence neuronal function and advance brain health through synergistic effects. Whole walnut extract guarded primary neuronal cells in vitro from oxidative and inflammatory stress caused by calcium dysregulation, which suppressed microglial activation [91 (Thangthaeng N, 2018)]. By raising the expression of neurotropic proteins like BDNF, which promote neuronal development and survival, n-3 PUFAs may also have neuroprotective benefits [92 (Hadjighassem M, 2015)]. Additional bioactive components in walnuts, including arginine, tocopherols, folate, melatonin, and polyphenols, promote cognitive health by modifying a number of cardiovascular risk factors [93 (E., 2015;)]. Almonds, Brazil nuts, and walnuts are the nuts with the highest concentration of free and total polyphenols[69 (Harnly JM, 2006;)].Pro-anthocyanidins, a kind of flavan-3-ol flavonoid, make up the majority of the polyphenols. Walnuts are a major source of ellagic acid, a phenolic antioxidant also present in pomegranates, strawberries, and almonds. There is an urgent need for more research on other nuts and cognition in both cognitively healthy and cognitively impaired people. Future research should also take into account pairing nutrients that have been shown to have significant impacts on cognition, such as blueberries and walnuts [74,94 (Nones, 2011)) (Poulose SM, ;2014)]. Although significant protection from ARCD (age related cognitive decline ) was not observed, older [96,95 (O’Brien J, 2014) (Koyama AK, 2015;) ] and middle-aged [97 (Nooyens AC, 2011;) ] persons who consumed nuts at least five times per week had higher global cognition and verbal memory composite scores than less frequent users. Younger and older persons who regularly ate walnuts had superior recall test scores, quicker reaction times, and higher single-digit learning scores than non-consumers in a cross-sectional study [98 (Arab L, 2015;)]. Similarly, consumption of legumes, nuts, and seeds was linked to better cognition in older adults from Greece, with regular users being less likely to receive subpar MMSE (Mini Mental State Examination) results [99 (Katsiardanis K, 2013;)]. Only two RCTs on nuts and cognition in the elderly, using either Brazil nuts or walnuts, have been conducted until present. Participants with MCI who ate one Brazil nut per day for six months performed better on tests of verbal fluency and constructional praxis but did not vary from controls in terms of global cognition [100 (Cardoso BR, 2016;) ]. Selenium, an antioxidant mineral used in seleno proteins crucial for brain function, is present in Brazil nuts [101 (Steinbrenner H, . 2013;)].
Cocoa
Flavonoids are abundant in cocoa, which is made from the dried, fermented seeds of the Theobroma cacao plant. The flavan-3-ol epicatechin, as well as catechin and other oligomeric procyanidins, are particularly abundant in cocoa. Epicatechins appear to pass the blood-brain barrier in animals and may act directly on the brain. Through improved neurogenesis, particularly the survival and proliferation of new hippocampal neurons, and an increase in synaptic development by BDNF stimulation, flavonoids from cocoa have been linked to improving neuroplasticity [66 ,73,72 (Gildawie KR, 2018;) (Krikorian R N. T.-H., 2010;)(Spagnuolo C, 2018)].
Moringa oleifera Lam.
The cruciferous plant Moringa oleifera Lam. (M. oleifera) is a member of the Moringaceae family. Locals refer to M. oleifera as the drumstick tree or the horseradish tree, and it is a widespread food worldwide. M. oleifera is used for both its nutritional and therapeutic qualities [102 (Mahmood, Mugal, & Haq, 2010, )]. Beta-carotene, vitamin C, vitamin E, and polyphenols are abundant in M. oleifera leaves, which are also a strong source of natural antioxidants [103 (Anwar, Latif, Ashraf, & Gilani, 2007,)]. A wide range of biological processes, including anti-inflammatory, anti-cancer, hepatoprotective, and neuroprotective properties, are now claimed to be improved by M. oleifera [102,105,104 (Mahmood, Mugal, & Haq, 2010, ) (Abdull, Ibrahim, & Kntayya, 2014, ) (Posmontier, 2011, )]. Numerous studies have also demonstrated its therapeutic benefits, including those for the treatment of diabetes, rheumatoid arthritis, atherosclerosis, infertility, pain alleviation, anti-depression, and thyroid and diuretic management [106, 107 (Banji, Banji, & Kavitha, 2012, ) (Chumark, et al., 2008, )]
Under challenging treatment circumstances, M. oleifera has been found to promote neuronal expansion and survival [109,108 (Hannan, et al., 2014, ) (Sutalangka, Wattanathorn, Muchimapura, & Thukham-mee, 2013,) ]. A 30 g/mL ethanol extract from M. oleifera leaves, for instance, has been shown to stimulate neurite outgrowth and neuronal differentiation from primary embryonic neurons in a concentration-dependent manner [108 (Hannan, et al., 2014, )]. The length of axons, the number and length of dendrites and axonal branching, and eventually the facilitation of synaptogenesis have all been seen to increase in response to M. oleifera leaf extract [108 (Hannan, et al., 2014, )]. Additionally, earlier research has shown that M. oleifera leaf extract effectively reduces neurodegeneration in hippocampal regions, and dentate gyrus leading to enhanced spatial memory [109 (Sutalangka, Wattanathorn, Muchimapura, & Thukham-mee, 2013,) ]. Mechanically, it can also lower levels of malondialdehyde (MDA) and acetylcholinesterase (AChE), while also boosting SOD and catalase (CAT) activity.
Piper
However, secondary metabolites from piper species have also demonstrated biological effects on human health. Piper species are aromatic plants used as culinary spices. The fruits, seeds, leaves, branches, roots, and stems of these plants contain a wealth of essential oils. Others, like Piper nigrum, Piper betle, and Piper auritum, include quite complex suites of secondary metabolites, whereas certain Piper species have straightforward chemical profiles. Globally, Piper species have been used in traditional medicine to treat a variety of illnesses, including urological issues, skin, liver, and stomach problems, wound healing, and as antipyretic and anti-inflammatory drugs. Additionally, Piper species may be employed in food preservation as natural antioxidants and antimicrobials.Compared to synthetic antioxidants, the phytochemicals and essential oils of Piper species have proven considerable antioxidant activity. They also showed antibacterial and antifungal properties against human infections. In addition, there are various chronic illnesses that Piper species can treat or prevent. Based on preclinical in vitro and in vivo studies, as well as clinical studies, it is believed that among the functional properties of Piper plants, extracts, and active components, the antiproliferative, anti-inflammatory, and neuropharmacological activities of the extracts and extract-derived bioactive constituents are key effects for the protection against chronic conditions.
The leading causes of disability and death worldwide are chronic diseases such type II diabetes, cardiovascular diseases, cancer, and neurological disorders. However, lifestyle adjustments, especially dietary changes, can prevent or at least delay the onset of the majority of these disorders[110 (Sharifi-Rad, et al., 2018, )]. It has been proven that Piper species have the ability to treat and prevent a number of chronic diseases. The neuropharmacological activities of the extracts and the bioactive elements produced from the extracts are regarded to be important impacts for the protection against chronic diseases, among the functional qualities of Piper plants.
Piperine has been shown to have significant effects on Central nervous system degenerative diseases including AD and PD. However, a number of pharmacological issues, including limited water solubility, significant first-pass metabolism, and low absolute oral bioavailability, make it difficult to transport piperine to the brain orally [111 (Elnaggar, Etman, Abdelmonsif, & Abdallah, Novel piperine-loaded Tween-integrated monoolein cubosomes as brain-targeted oral nanomedicine in Alzheimer’s disease: Pharmaceutical, biological, and toxicological studies. , 2015, )]. Recently, there has been a lot of research into the use of brain-targeted nanosystems to enhance the transport properties of difficult lipophilic active substances. Different solid lipid nanoparticles have been used in studies to demonstrate piperine encapsulation, and the results suggest that piperine-loaded nanoparticles significantly lessen the severity of neurodegenerative illnesses modeled in experimental animals [111,112 (Elnaggar, Etman, Abdelmonsif, & Abdallah, Novel piperine-loaded Tween-integrated monoolein cubosomes as brain-targeted oral nanomedicine in Alzheimer’s disease: Pharmaceutical, biological, and toxicological studies. , 2015, ) (Elnaggar, Etman, Abdelmonsif, & Abdallah, Intranasal piperine-loaded chitosan nanoparticles as brain-targeted therapy in Alzheimer’s disease: Optimization, biological efficacy, and potential toxicity. , 2015, )].In a recent study by Etman et al. [113 (Etman, Elnaggar, Abdelmonsif, & Abdallah, 2018, 19, )], piperine-loaded oral microemulsion as a nanosystem improved piperine efficacy and enhanced its delivery to the brain, leading to a better therapeutic outcome compared to the free drug in Alzheimer’s model male Wistar rats. Piperine-loaded chitosan-sodium tripolyphosphate nanoparticles improve neuroprotection and reduce astrocyte activation in a chemical kindling model of epilepsy, according to a recent study by Anissian et al. [114 (Anissian, et al., 2018,) ].
Cultural differences
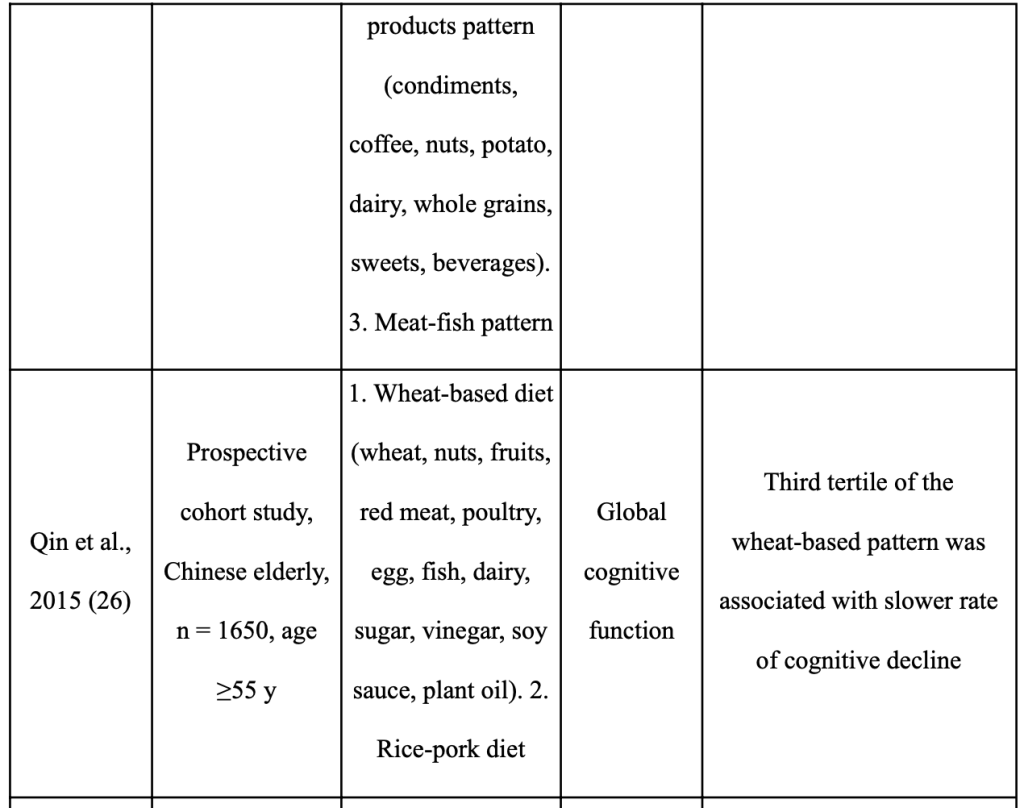
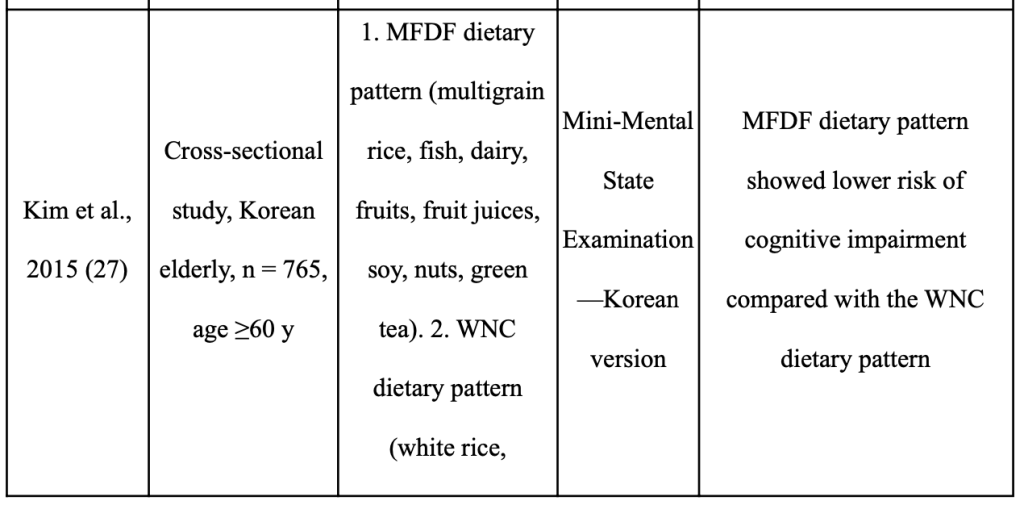
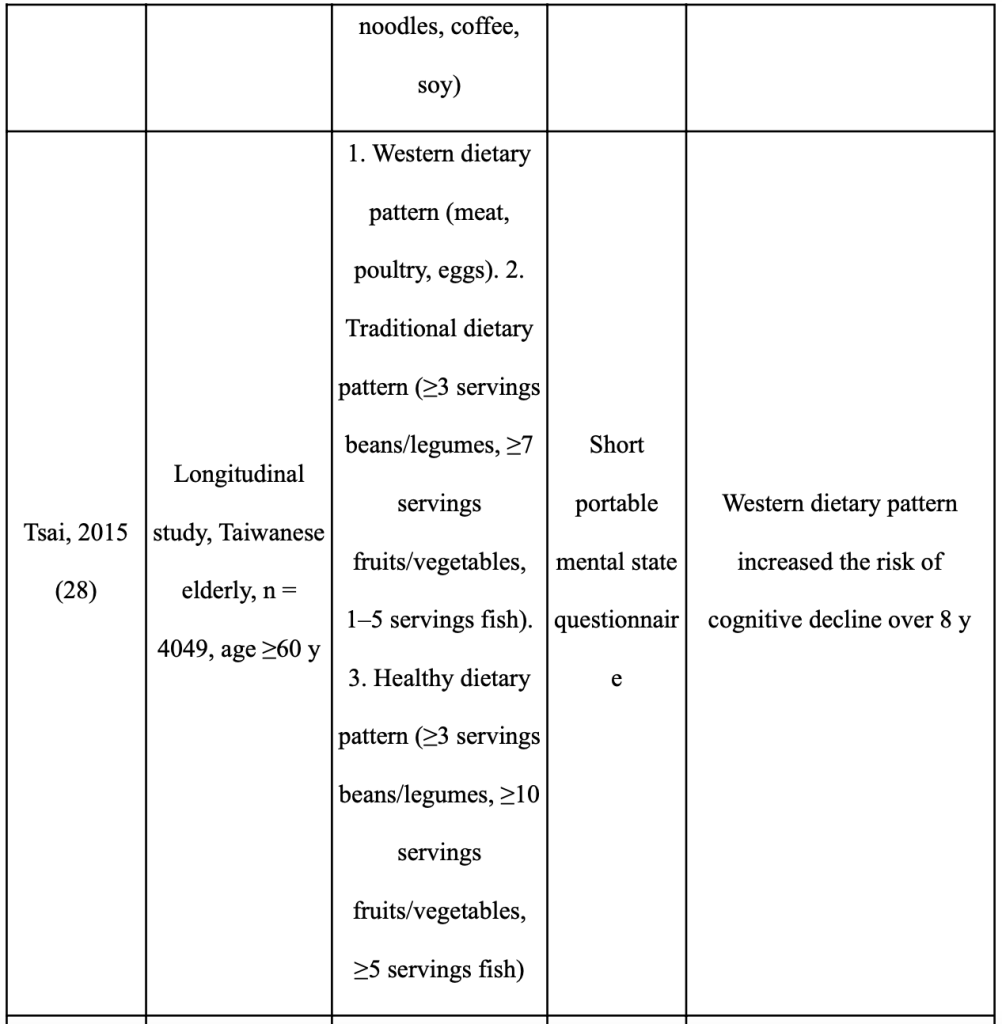
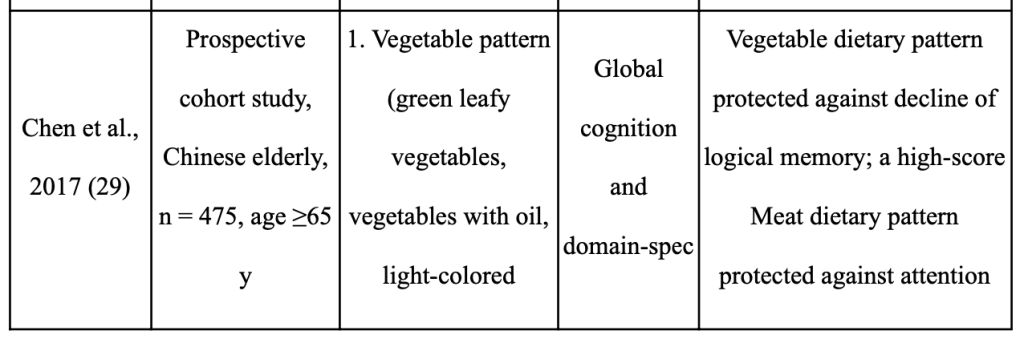
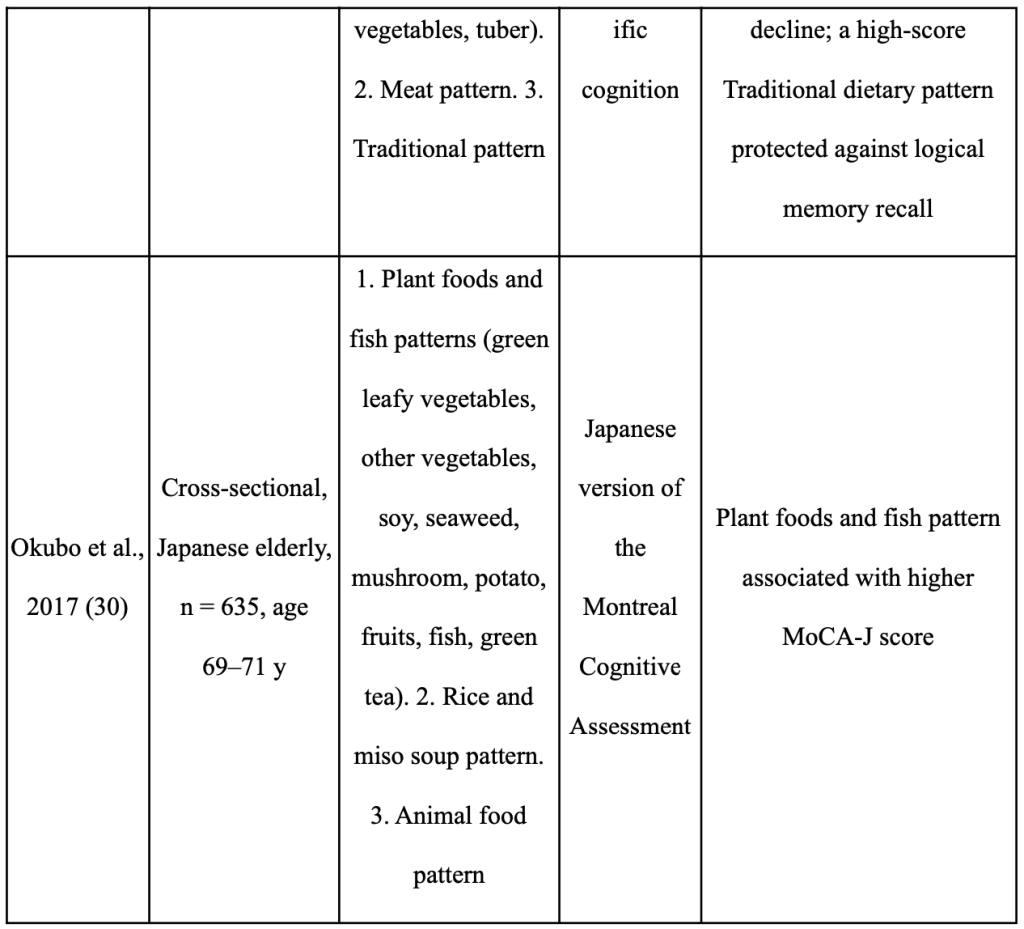
There is growing evidence that adopting a plant-based diet can reduce your risk of developing dementia and ARCD. The plant foods and nutrients prioritized in the Mediterranean, DASH, and MIND dietary patterns may be significant in preventing cognitive impairment, according to recent assessments of epidemiological data and intervention trials [116,117, 118,115, 119–124 (Abbatecola AM, 2018;) (Masana MF, 2017;) (Yannakoulia M, 2015) (Petersson SD, 2016;) (Chen X, 2019;) (Loughrey DG, 2017;) (Berendsen AM, 2018) (Berendsen AAM, 2017;) (Aridi YS, 2017;) (Morris MC, 2015;) ]. Despite the fact that these three dietary patterns include a diversity of plant foods, the dietary patterns of geographically distinct cohorts also show similarities in the prominence of particular plant foods and their overall positive effect on cognition [125–133 (Chan R, 2014;) (Qin B, 2015;) (Kim J, 2015;) (HJ., 2015) (Chen YC, 2017) (Okubo H, 2017) (Nurke E, 2010;) ]. These findings essentially agree with studies looking at the DII(Dietary Inflammatory Index) in connection to cognitive function [141, 134–137 (Frith E, 2018) (Shin D, 2018) (Hayden KM, 2017;) (Kesse-Guyot E, 2017;) (Assman KE, 2018.)].
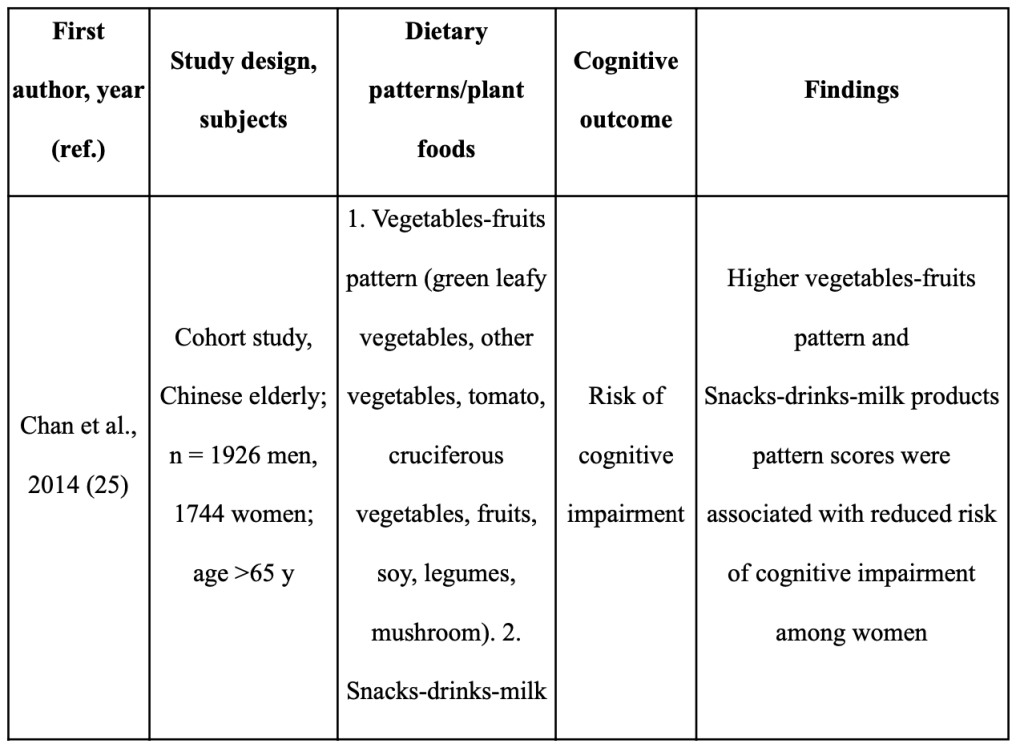
Table 5 summarizes the relationship between dietary habits that emphasize plants and cognition in a number of Asian cohorts. Green leaves and other vegetables, soy, whole grains, green tea, mushrooms, and seaweed are plant foods that Asians frequently eat [125–130 (Chan R, 2014;) (Qin B, 2015;) (Kim J, 2015;) (HJ., 2015) (Chen YC, 2017) (Okubo H, 2017) ]. Many of these plant-based diets are linked to lower rates of cognitive impairment [125, 126 (Chan R, 2014;) (Qin B, 2015;)], slower rates of cognitive decline [127,128 (Kim J, 2015;) (HJ., 2015)], better logical memory test results [129 (Chen YC, 2017)], and higher global cognitive assessment scores [130 (Okubo H, 2017)]. The cognitive outcomes of older persons from other nations (Norway, Australia, Italy) who consume a diet rich in fruits, vegetables, and legumes are likewise better [131–133 (Nurke E, 2010;) (Ashby-Mitchell K, 2015;) (Mazza E, 2017) ]. It’s interesting to note that Asian diets place less emphasis on the vibrant fruits, vegetables, nuts, non-soy legumes, and olive oil found in the Mediterranean diet and other dietary patterns. Techniques used in food preparation and processing are also crucial factors. The diets associated with better cognitive outcomes in various Asian cohorts [126, 129 (Qin B, 2015;) (Chen YC, 2017)] include stir-fried (in oil) and fermented vegetables (pickled cabbage), which may increase the bioavailability of phytochemicals [139,138 (Wickzkowski W, 2016;) (Bohn T, 2015) ]. Similar to this, it has been demonstrated that eating “multigrain rice” (brown rice, millets, black rice, and barley) rather than “white rice and noodles” reduces the risk of cognitive impairment in older Asians [126 (Qin B, 2015;) ], which is linked to the higher total polyphenol content in whole grains [140 (Goufo P, 2017;)]. Therefore, rather than relying on a single diet, the presence of a range of plant foods rich in accessible bioactive chemicals may be crucial for preventing ARCD [115–124 (Abbatecola AM, 2018;) (Masana MF, 2017;) (Yannakoulia M, 2015) (Petersson SD, 2016;) (Chen X, 2019;) (Loughrey DG, 2017;) (Berendsen AAM, 2017;) (Berendsen AM, 2018) (Aridi YS, 2017;) (Morris MC, 2015;) (Chan R, 2014;) ].
Both European Americans and African Americans have serum levels of 25(OH)D (25-hydroxyvitamin D) that are significantly linked with bone mineral density [142 (Bischoff-Ferrari, Dietrich, Orav, & Dawson-Hughes, 2004, )], but it has long been known that Africans and African Americans have higher bone mineral density (BMD) [143 ( Bell, 1997, )] and a lower risk of fragility fractures than Europeans [144 (Aloia, 2008, )]. Possible explanations include the fact that compared to European Americans, African Americans have higher calcium retention, poorer calcium outflow, and higher bone parathyroid hormone resistance [143,146,145 (Bell, 1997, ) (Aloia, 2008, ) (Gutierrez, Farwell, Kermah, & Taylor, 2011, )]. Although the cause of populations moving from Africa to higher latitudes evolving thinner bones is unknown, a trade-off for a decrease in pelvic deformity and labor obstruction has been proposed [147 (Vieth, 2020, )]. Whatever the causes, it appears that many people do not consider the low blood levels of 25(OH)D in African Americans to be a problem because of their stronger bones and the perception that the only effect of low 25(OH)D levels is poor bone health. Notably, the 2011 IOM review of vitamin D neglected to highlight how common vitamin D deficiency is among African Americans, even by their strictest definition of less than 12 ng/mL, and came to the conclusion that “requirements are being met by most, if not all, persons in both countries [US and Canada]”.
Food Preparation
Intermittent fasting and probiotic supplements both have positive effects on health on their own, but combining the two may increase these advantages. The PRObiotics and Intermittent FASTing to Improve Prediabetes (PROFAST) experiment sought to determine whether daily probiotic supplements and intermittent fasting can lower HbA1c in people with prediabetes. The findings of this study will add to the expanding body of research on the benefits of probiotic supplementation and intermittent fasting for the prevention of various diseases. As with other programmes, success depends on how closely participants follow dietary recommendations. Between their pre-intervention diet and their post-intervention diet, participants reported a moderate change in their diet. Additionally, participants noted that they were generally more conscious of their eating habits. Participants reported fewer episodes of overeating during the intervention, despite the fact that pretreatment binge eating disorder was not examined. This suggests that implementing an intermittent fasting regimen may affect eating behavior more favorably than unfavorably. Inconsistent correlations were discovered between the initiation of binge eating in people without previous binge eating and extreme dietary energy restriction (i.e., low or very low energy diets) in a systematic study that was published in 2015 [148 (Da Luz, et al., 2015, ) ]. As a result, it is confirmed that calorie restriction may not always result in unfavorable eating habits in those who do not have binge eating disorder, while preoperative screening is still advisable.[149 (Tay, (2020).)]
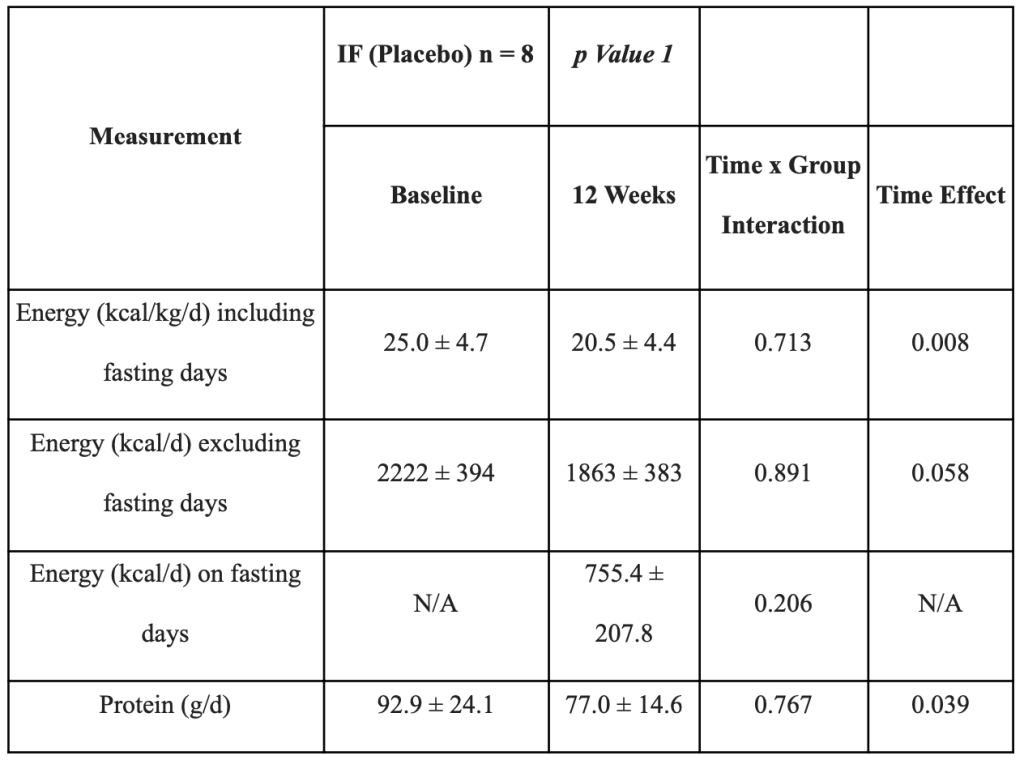
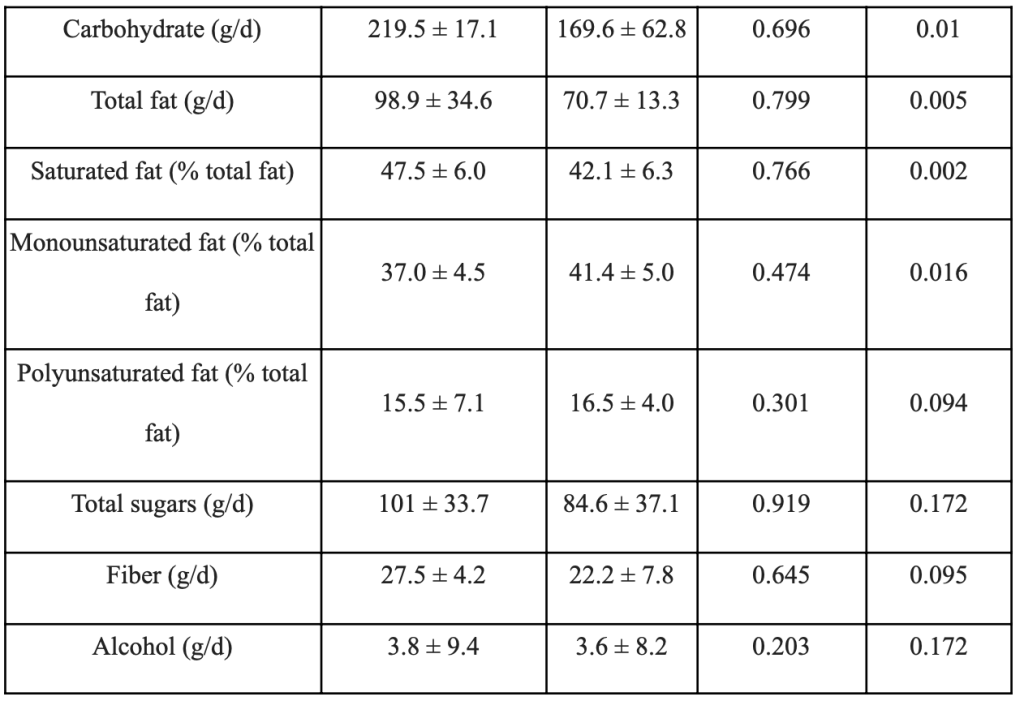
Data presented as mean ± standard deviation. Abbreviations: IF, intermittent fasting; g/d, grams per day; % TE, percentage of total energy. 1 Repeated measures analysis of variance. All nutrient measures include fasting days unless otherwise specified.
Conclusion
Evidence overwhelmingly points to plant-based dietary patterns that are rich in fruits, vegetables, nuts, seeds, legumes, and whole grains while placing less of an emphasis on processed foods and animal products as a desirable and effective strategy for preventing chronic diseases [150–152 (Carlos S, 2018;) (Medina-Remon A, 2018;) (Dinu M, 2017;)]. Such dietary patterns, from plant-exclusive diets to plant-centered diets, are linked to better long-term health outcomes and a lower risk of all-cause mortality [154, 153 (Kim H, 2018;) (Papandreou C, 2018;) ]. It is rational to assume that plant-based diets can reduce cognitive decline given that neurodegenerative illnesses and cardiovascular disorders share many pathophysiological pathways, such as oxidative stress, inflammation, and vascular damage.
Furthermore, Piper species have demonstrated their biological effects in numerous in vitro, in vivo, and clinical studies, in addition to the previously known usage in traditional medicine. Globally, the leading causes of disability and death are chronic illnesses and neurodegenerative diseases. Due to its antiproliferative, anti-inflammatory, and neuropharmacological properties, piper species have shown to have the ability to treat and prevent a number of chronic diseases. According to all of the research done on Piper species, these plants have potential as a cure for disorders that are inflammatory in nature. Due to the extensive use of standardized Piper plants, additional efforts should be made to research them using well-designed studies. The creation of functional meals based on Piper species also has a wide variety of potential applications.[155 (Salehi, (2019).)]
Additionally, Through the execution of its powerful anti-inflammatory action, inhibition of the activation of the NF-B and PI3K/Akt pathways, alleviating oxidative stress by scavenging free radicals, and improving neuroprotective roles, M. oleifera possesses a wide variety of medicinal and therapeutic qualities. M. oleifera can also control blood sugar levels and lower the chance of developing cancer, while the underlying mechanisms need more research. As a result, M. oleifera offers the possibility of preventing or treating a number of chronic disorders.
We should recognise that in addition to diet, lifestyle factors like getting enough sleep, relax, and exercise also affect the risk for cognitive impairment. The most efficient way to lower the population’s risk for age-related chronic decline ARCD may be a holistic strategy that addresses both dietary and lifestyle factors proven to enhance cognition. In conclusion, plant-centered eating habits that emphasize foods high in polyphenols, d-glutamate , herbs and nuts , fruits which can benefit the cognitive health of the aged population.[156 (Kou, (2018).)] Schizophrenia is a mental illness in which sufferers have psychotic symptoms such as delusions, hallucinations, and frequently disorganized speech. Additionally, patients may exhibit decreased emotional expressiveness, avolition, and impairment in social and vocational performance. Numerous investigations with people with schizophrenia revealed changes in the gut microbiota compared to healthy controls . Germ-free mice who received a faecal transplant from a person with schizophrenia had behaviors associated with schizophrenia [158,157 (Zheng P, 2019;) (Zhang X, 2020)]. In a human investigation, a decline in the number of Faecalibacterium can cause an increase in gut TH17 cells in schizophrenia patients [159 (Zhang X, 2020)]. These cells may pass the blood-brain barrier and stimulate the microglia in the hippocampus, resulting in aberrant behavior [159 (Zhang X, 2020)].
[Table 10]In addition to discovering that specific bacterial phyla like Veillonellaceae and Lachnospiraceae were connected to illness severity, Zheng et alstudy .’s on schizophrenia discovered that both untreated and treated individuals had changed gut microbiota [157 (Zheng P, 2019;)]. When compared to healthy controls, patients who had their first episode of psychosis had a change in their Lactobacillaceae [27Schwarz E, M. J. (2018;). Analysis of… ]. Additionally, this study discovered that during a one-year follow-up, people with the most changed microbiome populations had the lowest odds of illness remission. It’s interesting to see that Lactobacillaceae is on the rise because these bacteria are frequently found in probiotics and have been linked to anti-inflammatory and health-promoting benefits [160 (Naidu AS, 1999)]. According to human investigations, Lachnospiraceae, Akkermansia, and Sutterella levels were different in patients taking either risperidone or olanzapine compared to controls [161 (Flowers SA, 2017;)]. The scant evidence supporting probiotics’ effectiveness in treating schizophrenia was brought to light by a comprehensive study [162 (Ng Q, 2019)]. To fully comprehend the use of GM modification in the treatment of schizophrenia, more research will be required.
Discussion
Had we ever questioned why the world of emotions may revolve around food? Why do we spend so much time viewing culinary videos without even recognising it? Why do we often feel famished or have watery mouths and a calm energy when we see any delectable dish? How is new age culinary streaming gaining so much popularity? What could be the cause of individuals criticizing certain restaurants because they see other people eat or are dining in public? Either the person or the action you are watching tends to be in focus. If the second option is chosen, is watching others eat today so fascinating that people spend countless hours doing it?The interaction between the gut and the mind is crucial to consider when trying to understand human behavior like this. Food not only gives the body nourishment, but it also gives the brain the proper stimuli for a healthy mind. It’s a little strange to assume that food has the power to make or destroy both your intellect and you. One of our body’s most important and intelligent organs is the brain. If you don’t have control over your thoughts, they will begin to rule you. Therefore, it is also preferable to feed your brain in the manner in which you desire it to function; the simplest choice is food. Not only through consuming nutritious food but also by observing how it is prepared and presented. Does the person preparing your food put positive energy into it, which will then be absorbed by us when we eat it? Now you see why everyone simply adores their mother’s cooking—it is because, more than any of the ingredients, love and care have been transmitted through it. Almost everyone once in a lifetime encounters living alone, and it’s at such moments that we discover the effects cooking can have on us. It’s difficult to return home empty-handed after spending hours grocery shopping, especially if you only want to buy one item. To organize your groceries before returning home, to find your pantry full, and to feel content. Cooking your own food, plating it, sitting down to enjoy it, feeling accomplished, relieving yourself of all the day’s tension, and going to bed with a full stomach and a contented mind.
Similar to an engine consuming gasoline, our bodies function as machines thanks to the energy they receive from food. The food we consume for nutrition serves as a fuel for the mitochondria to produce energy. Oxygen and food-derived glucose interact to produce energy, carbon dioxide, and water molecules. Our body creates the energy needed to keep the heart beating, the lungs breathing, and the limbs moving by using the food we eat. In order to create muscle, we work out hard in the gym, which requires protein. Therefore, the advice from the gym instructors is to take those nutrients and eat a high-calorie diet. To achieve this goal, it is advised to consume protein-rich meals like lentils, milk, eggs, vitamin-rich vegetables, and meat in order to assist the body develop enough muscles.
Proteins in the meal offer amino acids for digestion. These amino acids play a range of activities, from assisting metabolism to safeguarding our heart, in the construction of proteins. The macronutrients that are essential for maintaining life, proteins help to form body tissues like cells and organs. When you are low on carbohydrates and fats, your body can also use amino acids as fuel. When we are ill, we take food and medicine to get better. If we eat enough food, our immunity increases and we experience a noticeable reduction in coughing and cold-like symptoms. Patients become extremely weak even in the majority of infections, ailments, or illnesses because they have stopped eating or are reluctant to do so.
Nutrition deficit is a type of illness where the disorder is brought on by either too little or too much food.
Inadequate nutrition combined with the use of medications causes a variety of issues, including drug buildup and the emergence of microbial drug resistance. This is due to the food’s ability to trigger enzymes. Drugs are thus made more water-soluble by metabolism and eliminated through urine. Food gives the body the strength it needs to safely metabolize drugs and medicines after they have taken effect in the body. Many oil-soluble medications are well absorbed when fatty foods . As frequently heard, the Korean recipe for beautiful skin, the Japanese secret of powerful muscles, etc. But have we ever questioned why such objective truths may be applied to a group as a whole and are almost universal? Something must be consumed by everyone and produce comparable outcomes for there to be such widespread similarity. For instance, a large portion of the Korean diet consists of rice, which contributes to their healthier skin and hair quality. This is now recognised globally, and eating rice in a similar manner has the desired effects. The prevalence of cognitive disorders in old age can be reduced by instilling strong morals in children and by educating them about the variety of foods available and the range of health benefits each one offers. Making it a habit, for instance, for your child to eat four soaked walnuts every day first thing in the morning may delay any age-related cognitive illnesses as well as any degeneration in the present.
We conclude by showing how various food categories, such as fruits, nuts, vegetables, beverages, and so forth, affect our cognitive behavior when consumed on a regular basis. The amount consumed depends on a variety of variables, including age, location, eating habits, how a particular food affects the brain-gut axis, and changes in gut bacteria. Additionally, the effect of nutrition might be seen as an environmental element in the development of SZ. A larger cohort study will be required in the future to investigate the various facets of food in the SZ spectrum.
Normal people who are approaching old age or who are in their 60s tend to have a box full of medications that they must take every day and are non-negotiable. Low energy and the sense of being unable to do anything can combine to make old age painful and not joyful as it would have been for someone before they reached their 60s. What would it feel like to be someone who has always been upbeat, healthy, and fine—smiling, having fun, and being the life of the party—instead? How would it make you feel? terrifying, huh? Why do some people experience that?simply old? Is being reliant on certain drugs to get through the day because of your age justified? No, absolutely not. Instead, make some lifestyle adjustments with one very crucial principle in mind: you should never skip meals, whether you’re feeling well or not. Simply being cautious and altering your lifestyle to give more importance to your eating habits—from choosing what to eat, incorporating important food types according to your body, to cooking and enjoying it—can make your life change in such a way that, regardless of your personal circumstances, you can always lead a high-quality life and maintain a positive outlook on life.Spreading and instilling in the next generation the value of learning about food consumption as a whole will increase their life expectancy as well as their quality of life, professional productivity, and ability to manage stress. A healthy gut also contributes to a healthy brain, which in turn improves overall health.
References
Abbatecola, A. M., Russo, M., & Barbieri, M. (2018). Dietary patterns and cognition in older persons. Current opinion in clinical nutrition and metabolic care, 21(1), 10–13. https://doi.org/10.1097/MCO.0000000000000434
Abdull, R., Ibrahim, M., & Kntayya, S. (2014, ). Health benefits of Moringa oleifera. . Asian Pac. J. Cancer Prev. APJCP , , 15, 8571–8576. .https://doi.org/10.7314/APJCP.2014.15.20.8571
Alagiakrishnan, T. H. ((2020) ). Gut microbes in neurocognitive and mental health disorders,. Annals of Medicine,, 52:8, 423-443.https://doi.org/10.1080/07853890.2020.1808239
Aloia, J. (2008, ). African Americans, 25-hydroxyvitamin D, and osteoporosis: A paradox. Am. J. Clin. Nutr. , 88, 545S–550S.https://doi.org/10.1007/s00109-011-0743-5
Anissian, D., Ghasemi-Kasman, M., Khalili-Fomeshi, M., Akbari, A., Hashemian, M., Kazemi, S., & Moghadamnia, A. (2018,). Piperine-loaded chitosan-STPP nanoparticles reduce neuronal loss and astrocytes activation in chemical kindling model of epilepsy. . Int. J. Biol. Macromol. , 107, 973–983.https://doi.org/10.1016/j.ijbiomac.2017.09.073
Anwar, F., Latif, S., Ashraf, M., & Gilani, A. ( 2007,). Moringa oleifera: A food plant with multiple medicinal uses. . . Phytother. Res., 21, 17–25.https://doi.org/10.1002/ptr.2023
Arab L, A. A. ( 2015;). A cross sectional study of the association between walnut consumption and cognitive function among adult US populations represented in NHANES. . J Nutr Health Aging., 19(3):284–90.https://doi.org/10.1007/s12603-014-0569-2
Aridi YS, W. J. (2017;). The association between the Mediterranean dietary pattern and cognitive health: a systematic review. . Nutrients. , 9:674. https://doi.org/10.3390/nu9070674
Ashby-Mitchell K, P. A. (2015;). Role of dietary pattern analysis in determining cognitive status in elderly Australian adults. . Nutrients. , 7:1052–67.https://doi.org/10.3390/nu7021052 Assman KE, A. M. (2018.). The inflammatory potential of the diet at midlife is associated with later healthy aging in French Adults. . J Nutr. ,;148:437–44.https://doi.org/10.1093/jn/nxx061
Bai, W., & Zhou, Y. (2017, ). Homeostasis of the Intraparenchymal-Blood Glutamate Concentration Gradient: Maintenance, Imbalance, and Regulation. Front. Mol. Neurosci. , 10, 400.https://www.frontiersin.org/articles/10.3389/fnmol.2017.00400/full
Baker, G., Prior, T., & Coutts, R. (2002,). Chirality and drugs used to treat psychiatric disorders. J. Psychiatry Neurosci. Jpn. , 27, 401–403.https://www.jpn.ca/content/jpn/27/6/401.full.pdf
Banji, O., Banji, D., & Kavitha, R. (2012, ). Immunomodulatory effects of alcbholic and hydroalcoholic extracts of Moringa olifera Lam. leaves. Indian J. Exp. Biol. , 50, 270–276. .https://pubmed.ncbi.nlm.nih.gov/22611915/
Bell, N. (1997, ). Bone and mineral metabolism in African Americans. . Trends Endocrinol. Metab. , 8, 240–245. .https://doi.org/10.1016/S1043-2760(97)00065-9
Bellono, N. W. ((2017)). Enterochromaffin Cells Are Gut Chemosensors that Couple to Sensory Neural Pathways. . Cell,, 170(1), 185–198.https://doi.org/10.1016/j.cell.2017.05.034
Bensalem J, D. S.-E. ( 2019;). Polyphenols from grape and blueberry improve episodic memory in healthy elderly with lower level of memory performance: a bicentric double blind, randomized, placebo-controlled-clinical study. J Gerontol A Biol Sci Med Sci., 74(7):996–1007. .https://doi.org/10.1093/gerona/gly166
Berendsen AAM, K. J. (2017;). The dietary approaches to stop hypertension diet, cognitive function, and cognitive decline in American older women. . J Am Med Dir Assoc. , 18(5):427–32.https://doi.org/10.1016/j.jamda.2016.11.026
Berendsen AM, K. J. (2018). Association of long-term adherence to the MIND diet with cognitive function and cognitive decline in American women. . J Nutr Health Aging. , ;22(2):222–9.https://doi.org/10.1007/s12603-017-0909-0
Bischoff-Ferrari, H., Dietrich, T., Orav, E., & Dawson-Hughes, B. ( 2004, ). Positive association between 25-hydroxy vitamin D levels and bone mineral density: A population-based study of younger and older adults. Am. J. Med., 116, 634–639.https://doi.org/10.1016/j.amjmed.2003.12.029
Bohmer, N., Dautel, A., Eisele, T., & Fischer, L. (2013, ). Recombinant expression, purification and characterisation of the native glutamate racemase from Lactobacillus plantarum NC8. . Protein. Expr. Purif. , 88, 54–60.https://www.sciencedirect.com/science/article/abs/pii/S104659281200321X
Bohn T, M. J.-M. ( 2015). Mind the gap-deficits in our knowledge of aspects impacting the bioavailability of phytochemicals and their metabolites—a position paper focusing on carotenoids and polyphenols. Mol Nutr Food Res., ;59(7):1307–23.https://doi.org/10.1002/mnfr.201400745
Bonaz BL, B. C. (2013). Brain-gut interactions in inflammatory bowel disease. Gastroenterology. , ;144(1):36–49.https://pubmed.ncbi.nlm.nih.gov/23063970/
Bookheimer SY, R. B. (2013;). Pomegranate juice augments memory and fMRI activity in middle-aged and older adults with mild memory complaints. Evid Based Complement Alternat Med., 2013:946298. .https://pubmed.ncbi.nlm.nih.gov/23970941/
Bowtell JL, A.-B. Z. (2017;). Enhanced task-related brain activation and resting perfusion in healthy older adults after chronic blueberry supplementation. Appl Physiol Nutr Metab. , 42(7):773–9.https://doi.org/10.1139/apnm-2016-0550
Brückner, H., & Hausch, M. (1989, ). Detectio0n of free D-amino acids in food by chiral phase capillary gas chromatography. . J. High Res. Chromatog. , 12, 680–684. .https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/jhrc.12401210 12
Brückner, H., & Hausch, M. (1990, ). D-amino acids in dairy products: Detection, origin and nutritional aspects. I. Milk, fermented milk, fresh cheese and acid curd cheese. . Milchwissenschaft . , 45, 357–360.https://www.cabdirect.org/cabdirect/abstract/19900441058
Burkholder-Cooley N, R. S.-S. ( 2016;). Comparison of polyphenol intakes according to distinct patterns and food sources in the Adventist Health Study-2 cohort. . Br J Nutr., 115(12):2126–9.doi:10.1017/S0007114516001331
Burrin, D., & Stoll, B. ( 2009, ). Metabolic fate and function of dietary glutamate in the gut. Am. J. Clin. Nutr, 90, 850S–856S. https://doi.org/10.3945/ajcn.2009.27462Y
Cani, P. D. ((2013)). Gut microbiota, enteroendocrine functions and metabolism. Current opinion in pharmacology,, 13(6), https://doi.org/10.1016/j.coph.2013.09.008
Caputi, V. &. (2018). Microbiome-gut-brain axis and toll-like receptors in parkinson’s disease. . Int. J. Mol. Sci. 19.https://doi.org/10.3390/ijms19061689 (2018).
Carabotti, M. S. (2015). Carabotti, M., Scirocco, A., Maselli, M. A., & Severi, C. . CAnnals of gastroenterology, , 28(2), 203–209.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4367209/
Cardoso BR, A. D.-F. ( 2016;). Effects of Brazil nut consumption on selenium status and cognitive performance in older adults with mild cognitive impairment: a randomized controlled pilot . Eur J Nutr., 55:107–16.https://doi.org/10.1007/s00394-014-0829-2
Carlos S, d. L.-A.-R.-C.-G.-C. ( 2018;). Mediterranean diet and health outcomes in the SUN cohort. Nutrients., 10(4):E439. https://doi.org/10.3390/nu10040439
Cattaneo, A. e. (2017). Association of brain amyloidosis with pro-inflammatory gut bacterial taxa and peripheral inflammation markers in cognitively impaired elderly. Neurobiol. Aging 49, , 60–68.https://doi.org/10.1016/j.neurobiolaging.2016.08.019
Cepeda, M. S. (2017). Microbiome-Gut-Brain Axis: Probiotics and Their Association With Depression. The Journal of neuropsychiatry and clinical neurosciences, 29(1), 39–44.https://doi.org/10.1176/appi.neuropsych.15120410
Chan R, C. D. (2014;). A cross-sectional study to examine the association between dietary patterns and cognitive impairment in older Chinese people in Hong-Kong. . J Nutr Health Aging. , 17:757–65. https://doi.org/10.1007/s12603-013-0348-5
Chang, C.-H. L.-H.-Y. ((2020)). d-glutamate and Gut Microbiota in Alzheimer’s Disease. . International Journal of Molecular Sciences,, 21(8), 2676. https://doi.org/10.3390/ijms21082676
Chen X, M. B. (2019;). Dietary patterns and cognitive health in older adults: a systematic review. . J Alzheimers Dis., 67(2):583–619.https://doi.org/10.3233/JAD-180468
Chen YC, J. C. (2017). Association of dietary patterns with global and domain specific cognitive decline in Chinese elderly. . J Am Geriatric Soc. , ;65(6):1159–67. https://doi.org/10.1111/jgs.14741
Chumark, P., Khunawat, P., Sanvarinda, Y., Phornchirasilp, S., Morales, N., Phivthong-Ngam, L., . . . Pongrapeeporn, K. (2008, ). The in vitro and ex vivo antioxidant properties, hypolipidaemic and antiatherosclerotic activities of water extract of Moringa oleifera Lam. leaves. . .J Ethnopharmacol. , 116, 439–446.https://doi.org/10.1016/j.jep.2007.12.010
Cohen, L. J.-F. (2017). Commensal bacteria make GPCR ligands that mimic human signalling molecules. . Nature, . , 549(7670), 48–53.https://doi.org/10.1038/nature23874
Da Luz, F., Hay, P., Gibson, A., Touyz, S., Swinbourne, J., Roekenes, J., & Sainsbury, A. (2015, ). Does severe dietary energy restriction increase binge eating in overweight or obese individuals? A systematic review. Obes. Rev. , 16, 652–665.https://doi.org/10.1111/obr.12295
Desbonnet, L. C. (2014). Microbiota is essential for social development in the mouse. . Molecular psychiatry,, 19(2), 146–148.https://doi.org/10.1038/mp.2013.65
Devore EE, K. J. (2012). Dietary intake of berries and flavonoids in relation to cognitive decline. Ann Neurol. , ;72(1):135–43. .https://doi.org/10.1002/ana.23594
Dinu M, A. R. (2017;). Vegetarian, vegan diets and multiple health outcomes: a systematic review with meta-analysis of observational studies. . Crit Rev Food Sci Nutr. , 57(17):3640–9.https://doi.org/10.1080/10408398.2016.1138447
Dyakin, V., Justin Lucas Dyakina-Fagnano, N., Posner, E., & Vadasz, C. (2017, ). The Chain of Chirality Transfer as Determinant of Brain Functional Laterality. Breaking the Chirality Silence: Search for New Generation of Biomarkers; Relevance to Neurodegenerative Diseases, Cognitive Psychology, and Nutrition Science. Neurol. Neurosci. Res. , 1, 2.
E., R. (2015;). Nuts and CVD. . Br J Nutr. , S2:S111–20.https://doi.org/10.3945/jn.113.184838
Elnaggar, Y., Etman, S., Abdelmonsif, D., & Abdallah, O. (2015, ). Intranasal piperine-loaded chitosan nanoparticles as brain-targeted therapy in Alzheimer’s disease: Optimization, biological efficacy, and potential toxicity. . J. Pharm. Sci. , 104, 3544–3556.https://doi.org/10.1002/jps.24557
Elnaggar, Y., Etman, S., Abdelmonsif, D., & Abdallah, O. (2015, ). Novel piperine-loaded Tween-integrated monoolein cubosomes as brain-targeted oral nanomedicine in Alzheimer’s disease: Pharmaceutical, biological, and toxicological studies. . Int. J. Nanomed., 10, 5459–5473. .https://doi.org/10.2147/IJN.S87336
Etman, S., Elnaggar, Y., Abdelmonsif, D., & Abdallah, O. (2018, 19, ). Oral brain-targeted microemulsion for enhanced piperine delivery in Alzheimer’s disease therapy: In vitro appraisal, in vivo activity, and nanotoxicity. . AAPS PharmSciTech , 3698–3711. https://doi.org/10.1208/s12249-018-1180-3
Flanagan E, M. M. (2018;.). Impact of flavonoids on cellular and molecular mechanisms underlying age-related cognitive decline and neurodegeneration. Curr Nutr Rep. , 7(2):49–57.https://doi.org/10.1007/s13668-018-0226-1
Flowers SA, E. S. (2017;). Interaction between atypical antipsychotics and the gut microbiome in a bipolar disease cohort. . Pharmacotherapy. , 37(3):261–267.https://doi.org/10.1002/phar.1890
Fotiadis, D., Kanai, Y., & Palacin, M. (2013, ). The SLC3 and SLC7 families of amino acid transporters. Mol. Asp. Med. , 34, 139–158.https://www.sciencedirect.com/science/article/abs/pii/S0098299712001215
Friebe A, D. A. (2011). Neutralization of LPS or blockage of TLR4 signaling prevents stress-triggered fetal loss in murine pregnancy. J Mol Med. , ;89(7):689–699https://doi.org/10.1007/s00109-011-0743-5
Frith E, S. N. (2018). Dietary inflammatory index and memory function: population-based national sample of elderly Americans. . Br J Nutr. ., ;119(5):552–8.https://doi.org/10.1017/S0007114517003804
Galli RL, C. A. ( 2016). Red raspberries can improve motor function in aged rats. J Berry Res., ;6:97–101.https://content.iospress.com/articles/journal-of-berry-research/jbr119
Gerald J Holtmann, A. C. (2016). Pathophysiology of irritable bowel syndrome. The Lancet Gastroenterology and Hepatology.https://doi.org/10.1016/S2468-1253(16)30023-1
Gildawie KR, G. R.-H. (2018;). Protective effects of foods containing flavonoids on age-related cognitive decline. Curr Nutr Rep. , 7:39–48.https://pubmed.ncbi.nlm.nih.gov/29892789/
Goufo P, T. H. ( 2017;). Factors influencing antioxidant compounds in rice. . Crit Rev Food Sci Nutr., 57(5):893–922.https://doi.org/10.1080/10408398.2014.922046
Gribble, F. M. (2016). Enteroendocrine Cells: Chemosensors in the Intestinal Epithelium. . Annual review of physiology,, 78, 277–299.https://doi.org/10.1146/annurev-physiol-021115-105439
Gutierrez, O., Farwell, W., Kermah, D., & Taylor, E. (2011, ). Racial differences in the relationship between vitamin D, bone mineral density, and parathyroid hormone in the National Health and Nutrition Examination Survey. . Osteoporos. Int. , 22, 1745–1753. https://doi.org/10.1007/s00198-010-1383-2
Hadjighassem M, K. B. ( 2015). Oral consumption of α-linolenic acid increases serum BDNF levels in healthy adult humans. . Nutr J. , 14:20.https://doi.org/10.1016/j.nutres.2017.10.016
Haller S, M. M.-L. (2018;). Impact of coffee, wine, and chocolate consumption on cognitive outcome and MRI parameters in old age. . Nutrients. , 10(10):E1391.https://doi.org/10.3390/nu10101391
Hannan, M., Kang, J., Mohibbullah, M., Hong, Y., Lee, H., Choi, J., . . . Moon, I. (2014, ). Moringa oleifera with promising neuronal survival and neurite outgrowth promoting potentials. . J. Ethnopharmacol. , 152, 142–150. .https://doi.org/10.1016/j.jep.2013.12.036
Harnly JM, D. R. (2006;). Flavonoid content of U.S. fruits, vegetables, and nuts. J Agric Food Chem. , 54(26):9966–77.https://pubmed.ncbi.nlm.nih.gov/17177529/
Hawkins, R., & Vina, J. (2016, ). How Glutamate Is Managed by the Blood-Brain Barrier. Biology , 5, 37.https://academic.oup.com/ajcn/article/90/3/867S/4597370
Hayden KM, B. D.-B. (2017;). The association between inflammatory diet and global cognitive function and incident dementia in older women: the women’s health initiative memory study. . Alzheimer’s Dement. , 13(11):1187–96.https://doi.org/10.1016/j.jalz.2017.04.004
Hays, S., Ordonez, J., Burrin, D., & Sunehag, A. (2007, ). Dietary glutamate is almost entirely removed in its first pass through the splanchnic bed in premature infants. Pediatr. Res. , 62, 353–356.https://www.nature.com/articles/pr2007231
Helms, H., Nielsen, C., Waagepetersen, H., & Brodin, B. (2017, ). Glutamate Transporters in the Blood-Brain Barrier. Adv. Neurobiol. , 16, 297–314. .https://link.springer.com/chapter/10.1007/978-3-319-55769-4_15
HJ., T. ( 2015). Dietary patterns and cognitive decline in Taiwanese aged 65 years and older. Int J Geriatr Psychiatry., ;5:523–30. https://doi.org/10.1002/gps.4176
Hu, S. e. (2019). Gut microbiota changes in patients with bipolar depression. Adv. Sci. 6, 1900752.https://doi.org/10.1002/advs.201900752 (2019).
J., D. G. (2013). Enteroendocrine cell signalling via the vagus nerve. Current opinion in pharmacology, , 13(6), 954–958.https://doi.org/10.1016/j.coph.2013.09.0077
J., D. G. (2014). Gastrointestinal hormones and the dialogue between gut and brain. The Journal of physiology, , 592(14), 2927–2941.https://doi.org/10.1113/jphysiol.2014.270850
Janeczko, M., Stoll, B., Chang, X., Guan, X., & Burrin, D. (2007, ). Extensive gut metabolism limits the intestinal absorption of excessive supplemental dietary glutamate loads in infant pigs. . J. Nutr. , 137, 2384–2390.https://doi.org/10.1093/jn/137.11.2384
Jiang H, L. Z. (2015). Altered fecal microbiota composition in patients with major depressive disorder. Brain Behav Immun. , ;48:186–194. .https://www.tandfonline.com/servlet/linkout?suffix=CIT0054&dbid=8&doi=10.1080%2 F07853890.2020.1808239&key=25882912
Jin, D., Miyahara, T., Oe, T., & Toyo’oka, T. (1999, ). Determination of D-amino acids labeled with fluorescent chiral reagents, R(−)- and S(+)-4-(3-isothiocyanatopyrrolidin-1-yl)-7-(N,N-dimethylaminosulfonyl)-2,1,3-benzoxad iazoles, in biological and food samples by liquid chromatography. Anal. Biochem. , 269, 124–132. .https://www.sciencedirect.com/science/article/abs/pii/S0003269798930901
Joris PJ, M. R. ( 2018). Cerebral blood flow measurements in adults: a review on the effects of dietary factors and exercise. . Nutrients., ;10(5):E530.https://doi.org/10.3390/nu10050530
Köhler CA, M. M. (2016;). The gut-brain axis, including the microbiome, leaky gut and bacterial translocation: mechanisms and pathophysiological role in Alzheimer’s disease. . Curr Pharm Des. , 22(40):6152–6166.https://pubmed.ncbi.nlm.nih.gov/27604604/
Katsiardanis K, D. A. (2013;). Cognitive impairment and dietary habits among elders: the Velestino Study. . J Med Food. , 16(4):343–50.https://doi.org/10.1089/jmf.2012.0225
Kawase, T., Nagasawa, M., Ikeda, H., Yasuo, S., Koga, Y., & Furuse, M. (2017, ). Kawase, T.; Nagasawa, M.; Ikeda, H.; Yasuo, S.; Koga, Y.; Furuse, M. . Br. J. Nutr. , 117, 775–783.https://www.cambridge.org/core/journals/british-journal-of-nutrition/article/gut- microbiota-of-mice-putatively-modifies-amino-acid-metabolism-in-the-host-brain/13EF1 2A7E949F8CBAA5286CB3117D30F
Kean RJ, L. D. (2015;1). Chronic consumption of flavanone-rich orange juice is associated with cognitive benefits: an 8-wk, randomized, double-blind, placebo-controlled trial in healthy older adults. . Am J Clin Nutr. , 01(3):506–14.https://pubmed.ncbi.nlm.nih.gov/25733635/
Kent K, C. K. (2017;). Food-based anthocyanin intake and cognitive outcomes in human intervention trials: a systematic review. . J Hum Nutr Diet. , 30:260–74.https://doi.org/10.1111/jhn.12431
Kesse-Guyot E, A. K. (2017;). Long-term association between dietary inflammatory index and cognitive functioning: findings from the SU.VI.MAX study. Eur J Nutr. , 56:1647–55.https://doi.org/10.1007/s00394-016-1211-3
Kim H, C. L. ( 2018;). Healthy plant-based diets are associated with lower risk of all-cause mortality in US adults. J Nutr., 148(4):624–31.https://doi.org/10.1093/jn/nxy019
Kim J, Y. A. ( 2015;). Dietary patterns derived by cluster analysis are associated with cognitive function among Korean older adults. Nutrients., 7(6):4154–69.https://doi.org/10.3390/nu7064154
Kobayashi, J. ( 2019, ). d-Amino Acids and Lactic Acid Bacteria. . Microorganisms , 7, 690.https://pubmed.ncbi.nlm.nih.gov/31842512/
Kou, X. L. ((2018).). Nutraceutical or Pharmacological Potential of Moringa oleifera Lam. . Nutrients, , 10(3), 343.https://doi.org/10.3390/nu10030343
Koyama AK, H. K. (2015;). Evaluation of a self-administered computerized cognitive battery in older population. . Neuroepidemiology. , 45(4):264–72.https://doi.org/10.1159/000439592
Krikorian R, N. T.-H. (2010;). Concord grape juice supplementation improves memory function in older adults with mild cognitive impairment. Br J Nutr. , 103(5):730–4.https://doi.org/10.1017/S0007114509992364
Krikorian R, S. M.-T.-H. (. 2010). Blueberry supplementation improves memory in older adults. . J Agric Food Chem. , ;58(7):3996–4000. .https://doi.org/10.1021/jf9029332
Lamport DJ, P. D. (2015;). The effect of flavanol-rich cocoa on cerebral perfusion in healthy older adults during conscious resting state: a placebo controlled, crossover, acute trial. Psychopharmacology (Berl). , 232(17):3227–34.https://doi.org/10.1093/jn/nxy019
Latorre, R. S.-V. (2016). Enteroendocrine cells: a review of their role in brain-gut communication. Neurogastroenterology and motility : the official journal of the European Gastrointestinal Motility Society, 28(5), 620–630.https://doi.org/10.1111/nmo.12754
Li, S. S. (2021). The gut microbiome is associated with brain structure and function in schizophrenia. . Sci Rep , 11, 9743 .https://doi.org/10.1038/s41598-021-89166-8
Li, S. Z. (2020). Altered gut microbiota associated with symptom severity in schizophrenia. PeerJ, , 8, e9574.https://doi.org/10.7717/peerj.9574
Lin, C., Yang, H., & Lane, H. (2019, ). D-glutamate, D-serine, and D-alanine differ in their roles in cognitive decline in patients with Alzheimer’s disease or mild cognitive impairment. . Pharm. Biochem. Behav. , 185, 172760. .https://www.sciencedirect.com/science/article/pii/S009130571930187X
Loughrey DG, L. S. (2017;). The impact of the Mediterranean diet on the cognitive functioning of older adults: a systematic review and meta-analysis. Adv Nutr. , 8:571–86.https://doi.org/10.3945/an.117.015495
Mahmood, K., Mugal, T., & Haq, I. (2010, ). Moringa oleifera: A natural gift—A review. J. Pharm. Sci. Res. , 2, 775–781. .https://scholar.google.com/scholar_lookup?title=Moringa+oleifera:+A+natural+gift%E2 %80%94A+review&author=Mahmood,+K.T.&author=Mugal,+T.&author=Haq,+I.U.&p ublication_year=2010&journal=J.+Pharm.+Sci.+Res.&volume=2&pages=775%E2%80% 93781
Mangas, A., Covenas, R., Bodet, D., Geffard, M., Aguilar, L., & Yajeya, J. ( 2007, ). Immunocytochemical visualization of d-glutamate in the rat brain. Neuroscience , 144, 654–664.https://www.sciencedirect.com/science/article/pii/S030645220601308X
Marchelli, R., Galaverna, G., Dossena, A., Palla, G., Bobbio, A., Santaguida, S., . . . Sforza, S. (2007; ). d-amino Acids: A New Frontier in Amino Acid and Protein Research; Konno, R., Bruckner, H., D’anillo, A., Fisher, G.H., Fujii, N., Homma, H., Eds. Eds.; Nova Science Publishers: New York, NY, USA, , pp. 299–315.https://www.sciencedirect.com/science/article/abs/pii/S0021967300914146
Marcone, G., Rosini, E., Crespi, E., & Pollegioni. (2020, ). L. D-amino acids in foods. . Appl. Microbiol. Biotechnol. , 104, 555–574.https://link.springer.com/article/10.1007/s00253-019-10264-9
Masana MF, K. A. (2017;). n-3 Fatty acids, Mediterranean diet and cognitive function in normal aging: a systematic review. . Exp Gerontol. , 91:39–50.https://doi.org/10.1016/j.exger.2017.02.008
Mazza E, F. A. ( 2017). Impact of legumes and plant proteins consumption on cognitive performances in the elderly. J Transl Med., ;15:109.https://doi.org/10.1186/s12967-017-1209-5
McNamara RK, K. W. (2018;). Cognitive response to fish oil, blueberry, and combined supplementation in older adults with subjective cognitive impairment. . Neurobiol Aging. , 64:147–56. .https://doi.org/10.1016/j.neurobiolaging.2017.12.003
Medina-Remon A, K. R.-R. (2018;). Dietary patterns and the risk of obesity, type-2 diabetes mellitus, cardiovascular diseases, asthma, and neurodegenerative diseases. . Crit Rev Food Sci Nutr. , 58(2):262–96.https://pubmed.ncbi.nlm.nih.gov/27127938/
Miller MG, H. D.-H. (2018;). Dietary blueberry improves cognition among older adults in a randomized, double-blind, placebo-controlled trial. . Eur J Nutr. , 57(3):1169–80https://doi.org/10.1007/s00394-017-1400-8
Morillo Sarto, H. B.-S.-M.-G.-C.-M. (2019). Efficacy of a mindful-eating programme to reduce emotional eating in patients suffering from overweight or obesity in primary care settings: a cluster-randomised trial protocol. BMJ open, 9(11).https://doi.org/10.1136/bmjopen-2019-031327
Morris MC, T. C. (2015;). MIND diet slows cognitive decline with aging. . Alzh Dement. , 11(9):1015–22.https://doi.org/10.1016/j.jalz.2015.04.011
Mothet, J., & Snyder, S. (2012, .). Brain d-amino acids: A novel class of neuromodulators. . Amino Acids , 43, 1809–1810.https://link.springer.com/article/10.1007/s00726-012-1403-y
Naidu AS, B. W. ( 1999). Probiotic spectra of lactic acid bacteria (LAB). . Crit Rev Food Sci Nutr., ;39(1):13–126.https://doi.org/10.1080/10408699991279187
Ng Q, S. A. (2019). A systematic review of the effect of probiotic supplementation on schizophrenia symptoms. . Neuropsychobiology. , ;78(1):1–6.https://doi.org/10.1159/000498862
Nguyen, T. T. (2018). Overview and systematic review of studies of microbiome in schizophrenia and bipolar disorder. . J. Psychiatr. Res. , 99, 50–61.https://doi.org/10.1016/j.jpsychires.2018.01.013 (2018).
Nguyen, T. T. (2019). Differences in gut microbiome composition between persons with chronic schizophrenia and healthy comparison subjects. ScienceDirect ELSEVIER.https://doi.org/10.1016/j.schres.
Nilsson A, S. I. (2017;). Effects of a mixed berry beverage on cognitive functions and cardio metabolic risk markers: a randomized cross-over study in healthy adults. PLoS One. , 12(11):e0188173.https://doi.org/10.1371/journal.pone.0188173
Nones, J. E. (2011)). . Hesperidin, a flavone glycoside, as mediator of neuronal survival. . Neurochemical research, , 36(10), 1776–1784..https://doi.org/10.1007/s11064-011-0493-33
Nooyens AC, B.-d.-M. H. (2011;). Fruit and vegetable intake and cognitive decline in middle-aged men and women: the Doetinchem cohort study. . Br J Nutr. , 106(5):752–61.https://doi.org/10.1017/S0007114511001024
Nurke E, R. H. (2010;). Cognitive performance among the elderly in relation to the intake of plant foods. The Hordaland Health Study. . Br J Nutr. , 104(8):1190–201.https://doi.org/10.1017/S0007114510001807
O’Brien J, O. O. (2014). Long-term intake of nuts in relation to cognitive function in older women. J Nutr Health Aging. , ;18(5):496–502.https://doi.org/10.1007/s12603-014-0014-6
Okubo H, I. H. (2017). . Association between dietary patterns and cognitive function among 70 year old Japanese elderly: a cross-sectional analysis of the SONIC study. Nutr J. , ;16(1):56.https://doi.org/10.1186/s12937-017-0273-2
Osadchiy, V. M. (2019). The Gut-Brain Axis and the Microbiome: Mechanisms and Clinical Implications. Clinical gastroenterology and hepatology . the official clinical practice journal of the American Gastroenterological Association, 17(2), 322–332.https://doi.org/10.1016/j.cgh.2018.10.002
Palla, G., Marchelli, R., Dossena, A., & Casnati, G. (1989, ). Occurrence of D-Vamino acids in food: Detection by capillary gas chromatography and by reversed-phase high-performance liquid chromatography with L-phenylalaninamides as chiral selectors. J. Chrom. A , 475, 45–53.https://www.sciencedirect.com/science/article/abs/pii/S0021967300914146
Papandreou C, B.-T. N.-G. (2018;). . Legume consumption and risk of all-cause, cardiovascular, and cancer mortality in the PREDIMED study. . Clin Nutr. , 38(1):348–56.https://doi.org/10.1016/j.clnu.2017.12.019
Petersson SD, P. E. (2016;). Mediterranean diet, cognitive function, and dementia: a systematic review of the evidence. . Adv Nutr. , 7:889–904.https://doi.org/10.3945/an.116.012138
Polito CA, C. Z.-Y.-L.-M.-C.-P.-R. (2018;). . Association of tea consumption with risk of Alzheimer’s disease and anti-beta amyloid effects of tea. . Nutrients. , 10(5):E655.https://doi.org/10.3390/nu10050655
Posmontier, B. (2011, ). The medicinal qualities of Moringa oleifera. . Holist. Nurs. Pract. , 25, 80–87. .https://doi.org/10.1097/HNP.0b013e31820dbb27
Potì, F. S. ((2019)). Polyphenol Health Effects on Cardiovascular and Neurodegenerative Disorders: A Review and Meta-Analysis. . International journal of molecular sciences, , 20(2), 351.https://doi.org/10.3390/ijms20020351
Poulose SM, M. M.-H. (;2014). Role of walnuts in maintaining brain health with age. . J Nutr., 144(4S):561S–6S.https://doi.org/10.3945/jn.113.184838
Psichas, A. R. ((2015).). Gut chemosensing mechanisms. . The Journal of clinical investigation, 1, 25(3), 908–917.. https://doi.org/10.1172/JCI76309
Qin B, A. L. (2015;). Dietary patterns and cognitive decline among Chinese older adults. Epidemiology. , 26(5):758–68.https://doi.org/10.1097/EDE.0000000000000338
Quddusi, M. A. (2018, 07 18). The Scientific World. Retrieved from https://www.scientificworldinfo.com/2018/07/importance-of-food-in-our-daily-life.htm
Redmond, J., Palla, L., Yan, L., Jarjou, L., Prentice, A., & Schoenmakers, I. (2015, ). Ethnic differences in urinary calcium and phosphate excretion between Gambian and British older adults. Osteoporos.Int. , 26, 1125–1135. https://doi.org/10.1007/s00198-014-2926-8
Reeds, P., Burrin, D., Stoll, B., & Jahoor, F. (2000, ). Intestinal glutamate metabolism. Nutr. , 130, 978S–982S.https://academic.oup.com/jn/article/130/4/978S/4686655
Rhee SH, P. C. (2009;). Principles and clinical implications of the brain-gut-enteric microbiota axis. Nat Rev Gastroenterol Hepatol. ., 6:306–314.[PMC free article] [PubMed] [Google Scholar]
Saha, S. C. (2007). A systematic review of mortality in schizophrenia: is the differential mortality gap worsening over time? Arch. Gen. Psychiatry, 64, 1123–1131https://doi.org/10.1001/archpsyc.64.10.1123 (2007).. .
Salehi, B. Z.-R. ( (2019).). Piper Species: A Comprehensive Review on Their Phytochemistry, Biological Activities and Applications. . Molecules, , 24(7), 1364http://dx.doi.org/10.3390/molecules24071364
Sampson, T. R.-B.-S. (2016). Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease. Cell, , 167(6), 1469–1480.e12.https://doi.org/10.1016/j.cell.2016.11.018
Schwarz E, M. J. (2018;). Analysis of microbiota in first episode psychosis identifies preliminary associations with symptom severity and treatment response. Schizophr Res. , 192:398–403. .https://doi.org/10.1016/j.schres.2017.04.017
Severance EG, G. K. (2013). Discordant patterns of bacterial translocation markers and implications for innate immune imbalances in schizophrenia. Schizophrenia Res. , ;148(1–3):130–137.https://pubmed.ncbi.nlm.nih.gov/23746484/
Sharifi-Rad, J., Tayeboon, G., Niknam, F., Sharifi-Rad, M., Mohajeri, M., Salehi, B., . . . Sharifi-Rad, M. V. (2018, ). extract—Antibacterial, antifungal and scolicidal activities, and inhibitory potential on acetylcholinesterase, tyrosinase, lipoxygenase and xanthine oxidase. . . Cell. Mol. Biol. , 64, 50–56.https://doi.org/10.1016/j.tifs.2018.08.008
Shin D, K. S. (2018). Inflammatory potential of diet is associated with cognitive function in older adult Korean population. . Nutrition. , ;55-56:56–62.https://doi.org/10.1016/j.nut.2018.02.026
Shriram, R. (22, 04 10). ACME COLLINS SCHOOL. Retrieved from https://www.acmecollinsschool.com/blog/importance-of-food-in-our-daily-lives
Shukkit-Hale B, B. D. (2015;). The beneficial effects of berries on cognition, motor behavior and neuronal function in aging. . Br J Nutr. , 114(10):1542–9.https://doi.org/10.1017/S0007114515003451
Simeonova D, I. M. (2018). Recognizing the leaky gut as a trans-diagnostic target for neuroimmune disorders using clinical chemistry and molecular immunology assays. C. Curr Top Med Chem. , ;18(19):1641–1655.https://www.tandfonline.com/servlet/linkout?suffix=CIT0072&dbid= 8&doi=10.1080%2F07853890.2020.1808239&key=30430944
Soken-Huberty, E. (21, 12 15). THE IMPORTANT SITE. Retrieved from https://theimportantsite.com/10-reasons-why-food-is-important/ https://theimportantsite.com/10-reasons-why-food-is-important/
Souza DG, V. A. ( 2004;). The essential role of the intestinal microbiota in facilitating acute inflammatory responses. J Immunol., 173(6):4137–4146.https://www.tandfonline.com/servlet/linkout?suffix=CIT0031&dbid=8&doi=10.1080%2F07853890.2020.1808239&key=15356164
Spagnuolo C, M. S. (2018). Anti-inflammatory effects of flavonoids in neurodegenerative disorders. Eur J Med Chem. , 153:105–15.https://doi.org/10.1016/j.ejmech.2017.09.001
Steinbrenner H, S. H. (. 2013;). Selenium homeostasis and antioxidant selenoproteins in brain:implications for disorders in the central nervous system. . Arch Biochem Biophys, 536(2):152–7. https://doi.org/10.1016/j.abb.2013.02.021
Sudo, N. e. (2004). Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. . J. Physiol.558, , 263–275.https://doi.org/10.1113/jphysiol.2004.063388(2004).
Sujatha Rajaram, J. J. (Issue Supplement_4, November 2019). Plant-Based Dietary Patterns, Plant Foods, and Age-Related Cognitive Decline, Advances in Nutrition. Volume 10, , , Pages S422–S436,.http://dx.doi.org/10.3390/molecules24071364
Sutalangka, C., Wattanathorn, J., Muchimapura, S., & Thukham-mee, W. (2013,). Moringa oleifera mitigates memory impairment and neurodegeneration in animal model of age-related dementia. . Oxid. Med. Cell. Longev. , 695936. .https://doi.org/10.1155/2013/695936
Tay, A. P. ((2020).). PROFAST: A Randomized Trial Assessing the Effects of Intermittent Fasting and Lacticaseibacillus rhamnosus Probiotic among People with Prediabetes. . Nutrients, , 12(11), 3530.https://doi.org/10.3390/nu12113530
Thangthaeng N, P. S.-H. ( 2018). Walnut extract modulates activation of microglia through alteration in intracellular calcium concentration. . Nutr Res., ;49:88–95.https://doi.org/10.1016/j.nutres.2017.10.016
Tomova, A. e. (2015). Gastrointestinal microbiota in children with autism in Slovakia. Physiol. Behav. , 138, 179–187.https://doi.org/10.1016/j.physbeh.2014.10.033 (2015)
Travica N, D. N. (2019, ). The effect of blueberry interventions on cognitive performance and mood: A systematic review of randomized controlled trials. . Brain Behav Immun. , pii: S0889-1591(18):31195-4.https://www.sciencedirect.com/science/article/abs/pii/S0889159 118311954
Valls-Pedret C, L.-R. R.-R. ( 2012). Polyphenol-rich foods in the Mediterranean diet are associated with better cognitive function in elderly subjects at high cardiovascular risk. J Alzheimers Dis., 29(4):773–82.https://pubmed.ncbi.nlm.nih.gov/22349682/
Vieth, R. (2020, ). Weaker bones and white skin as adaptions to improve anthropological “fitness” for northern environments. Osteoporos. Int. , 31, 617–624. .https://doi.org/10.1007/s00198-019-05167-4
Whyte AR, C. N. (. 2018;.). A randomized double blind placebo controlled study to compare the safety and efficacy of low dose enhanced wild blueberry powder and wild blueberry extract in maintenance of episodic and working memory in older adults. Nutrients, 10(6):E660.https://doi.org/10.3390/nu10060660
Wickzkowski W, S.-N. D. (2016;). The impact of red cabbage fermentation on bioavailability of anthocyanins and antioxidant capacity of human plasma. Food Chem. , 190:730–40.https://doi.org/10.1016/j.foodchem.2015.06.021
Yannakoulia M, K. M. ( 2015). Cognitive health and Mediterranean diet: just diet or lifestyle pattern? . Age Res Rev., ;20:74–8.https://doi.org/10.1016/j.arr.2014.10.003
Zamora-Ros R, K. V.-R. (2016;). Dietary polyphenol intake in Europe: the European Prospective Investigation into Cancer and Nutrition (EPIC) study. . Eur J Nutr. , 55(4):1359–75.https://doi.org/10.1007/s00394-015-0950-x
Zhang X, P. L. (2020). Analysis of gut mycobiota in first-episode, drug-naïve Chinese patients with schizophrenia: a pilot study. . Behav Brain Res. , ;379:112374. https://doi.org/10.1016/j.bbr.2019.112374
Zheng P, Z. B. (2019;). The gut microbiome from patients with schizophrenia modulates the glutamate-glutamine-GABA cycle and schizophrenia-relevant behaviors in mice. Sci Adv. , 5(2):eaau8317https://doi.org/10.1126/sciadv.aau8317
Zhu F, G. R. (2019.). Transplantation of microbiota from drug-free patients with schizophrenia causes schizophrenia-like abnormal behaviors and dysregulated kynurenine metabolism in mice. . Mol Psychiatry. https://doi.org/10.1038/s41380-019-0475-4
Figures
https://www.cghjournal.org/action/showFullTableHTML?isHtml=true&tableId=tbl1&pii=S1542-3565%2818%2931081-4
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4367209/figure/F1/ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4367209/
Tables
https://www.cghjournal.org/action/showFullTableHTML?isHtml=true&tableId=tbl1&pii=S1542 -3565%2818%2931081-4
https://www.mdpi.com/1422-0067/21/8/2676/htm#B55-ijms-21-02676
https://academic.oup.com/view-large/185745750
https://academic.oup.com/view-large/185745754
https://academic.oup.com/view-large/185745726
https://www.mdpi.com/2072-6643/12/11/3530
About the author

Kajal Gupta
Kajal has a strong love for research and is incredibly fascinated about various issues in the health sector that require answers in order to keep up with the expanding global population.
